Show all large animal articles
Managing Milk Fever in Dairy Cows
Sara Pedersen BSc(Hons) BVetMed CertCHP DBR MRCVS RCVS Specialist in Cattle Health and Production - 03/09/2017
Managing Milk Fever in Dairy Cows
The majority of clinical cases of milk fever are relatively straightforward and low-cost to treat, resulting in a large underestimation of the true impact the disorder has on both individual and herd productivity. Milk fever, or hypocalcaemia, is the most common metabolic disorder in cattle in the UK with an estimated annual incidence of 4-9% (Husband, 2005), although on some farms the clinical incidence can be as high as 50-60%. However, clinical cases may only be the tip of the iceberg as more cows are likely to be affected by subclinical milk fever.
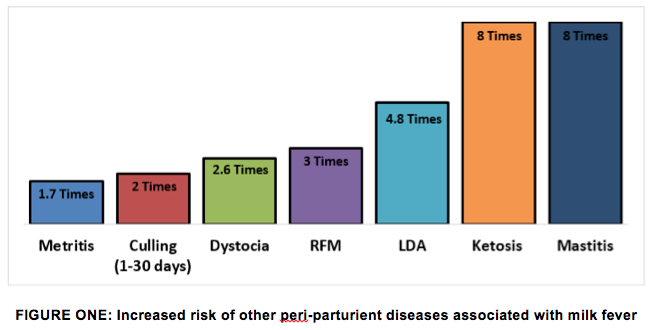
The average cost of a case of clinical milk fever is considerably higher than the cost of treatment. The most recent calculations give an average cost of £250 per case (Husband, 2011) with the majority of costs resulting from the knock on effects in terms of production disease. Cows with milk fever are more at risk of retained cleansing, mastitis, metritis, endometritis and left displaced abomasum (Houe et al., 2001; Figure One). Therefore as with many diseases and disorders, prevention is always more cost-effective than cure.
What is Milk Fever?

Milk fever is a non-nutritional, non-degenerative production disease that occurs predominantly around calving due to a sudden increase in calcium demand and an unavoidable delay in calcium metabolism adaptation. The redirection of calcium from the foetus to the udder results in a rapid increase in calcium clearance from the blood such that the majority of cows experience hypocalcaemia to some degree. A pregnant cow has a dietary requirement of 30g per day but a cow producing 40 litres of milk requires 80g per day and even more when producing colostrum.
Although the majority of cows will experience hypocalcaemia to some degree, risk does increase with both age and thus parity with a reported 9% increase with each lactation (DeGaris & Lean, 2008). In addition high yielding cows, those with a history of milk fever, a body condition score (BCS) >3.5, on a grass based diet or a diet high in potassium are also at greater risk (Husband, 2005).
For every case of clinical milk fever in the herd there are approximately 3-6 cases of subclinical milk fever (Houe et al., 2001). However, some herds can have a high incidence of subclinical milk fever without experiencing clinical cases. Subclinical milk fever is defined as a serum calcium level of <2mmol/L in the absence of clinical signs. Based on this threshold a study found that 25% of heifers, 41% of second lactation cows and up to 54% of fifth lactation cows had subclinical disease (Reinhardt et al., 2011).
Calcium Homeostasis
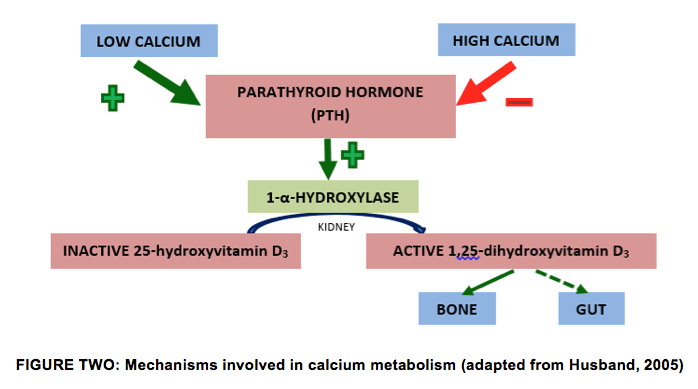
Serum calcium levels are regulated by hormonal signals, with the most important being parathyroid hormone (PTH) and 1,25-dihydroxyvitamin D3 (calcitriol), which mainly modulate renal calcium reabsorption, gastrointestinal calcium transport and bone calcium turnover.
Both Vitamin D2 and D3 are present in low levels in the blood and are converted to inactive 25-hydroxyvitamin D3 in the liver. This in turn is converted to the active form 1,25-dihydroxyvitamin D3 in the kidney by the enzyme 1-α-hydroxylase. PTH plays a role in the in the upregulation of 1-α-hydroxylase activity and thus 1,25-dihydroxyvitamin D3 production, resulting in increased absorption from the gut and resorption from the bone (Figure 2).
Increasing calcium and 1,25-dihydroxyvitamin D3 levels act via negative feedback to downregulate the process. Cows with milk fever have abnormal levels of, but a reduced sensitivity to the PTH and 1,25-dihydroxyvitamin D3 (Husband, 2005).
Although not directly involved in the regulation of calcium, magnesium is involved in the production and release of PTH, thus hypomagnesaemia can impact on calcium homeostasis indirectly.
Treatment
The majority of cases of milk fever can be treated relatively simply with the administration of calcium, however, recurrence occurs in around 1 in 3 cases (Thilsing-Hansen et al, 2002). The aim of treatment is to ‘bridge’ the gap whilst the cow adapts to the increase in calcium demand by mobilising her own reserves. Following intravenous (IV) administration blood calcium levels rise rapidly and will remain high for 5-6 hours, which is sufficient time to enable calcium metabolism and regulation to stabilise. Following subcutaneous administration it takes 3-4 hours to reach target levels and a quick response to treatment is less likely. By 12 hours all administered calcium will have been eliminated.
A 600kg cow needs 8-12g of calcium by slow IV injection as soon as possible after she starts to show signs of milk fever. A 400ml bottle of a 40% calcium solution e.g. Calciject CM40, contains 12g. IV administration is also preferable because cows with milk fever do not have good peripheral circulation (hence the cold ears) so they do not absorb calcium well from under the skin. There is some evidence to suggest that giving two bottles in the vein is counterproductive and may slow down the adaptation process and increase the recurrence rate. Too much calcium can also weaken the heart muscles. 85% of cows respond to one treatment; many rise within 10 minutes and others 2-4 hours later.
Giving one bottle IV and another under the skin does not affect recurrence rate (25%) and can increase the likelihood of ‘downer cow syndrome’. Instead, providing additional oral supplementation of calcium in the form of a drench or bolus can reduce the risk of recurrence as it provides slow release of calcium whilst at the same time boosting the cow’s own ability to release calcium from the bones and increase uptake from the gut.
Prevention
If a farm is experiencing too many cases of clinical milk fever (>3%) or is found to have a high incidence of subclinical milk fever then a preventative strategy is required. There are a number of options which either target the herd or the individual cow.
Decreased Dietary Supply Before Calving:
Lowering dietary calcium in the close-up diet is very effective at preventing milk fever, especially when calcium intake can be lowered to 20g per day. However, in reality this is difficult to achieve alongside the requirement to reduce energy intake at this time. Forages are high in calcium and are a traditional component of dry cow rations thus lowering the calcium intakes to much less than 60g per day can be difficult. Whilst straw offers a low calcium substitute, its palatability limits intakes just at a time when maximising intakes is crucial to prevent excessive negative energy balance post-calving.
Another option is to feed a calcium antagonist or binder, such as a zeolite clay. Any calcium ingested is bound to the antagonist and thus unable to be absorbed so the cow is allowed to adapt metabolically and can rapidly mobilise calcium at calving. A disadvantage to calcium binders can be their impact on dry matter intakes although if fed at moderate doses (23g/kg DM) suppression of intakes does not compromise the effectiveness of the binder (Grabherr et al., 2009). Due to the cost of calcium binders this strategy is often restricted to farms that have separate ‘far-off’ and ‘close-to’ groups of dry cows rather than a single group so that the binder is only fed in the last few weeks prior to calving.
Reduction of Dietary Cation-Anion Difference:

This is a commonly adopted nutritional preventive strategy that can be an extremely effective way of controlling milk fever if implemented correctly. It is based on inducing a moderate metabolic acidosis by the modification of the dietary DCAD. This is achieved by feeding mineral supplements that contain sulphur or chlorine, without sodium or potassium, commonly referred to as anionic salts. Induction of metabolic acidosis upregulates the action of PTH on 1-α-hydroxylase as well as the effect of 1,25-dihydroxyvitamin D3 on resorption of calcium from the bone and uptake from the gut, although it has a greater effect on the former.
Feeding a full DCAD ration requires a thorough and strategic approach, thus it is not an advisable strategy for every farm. The correct balance of anionic salts must be fed, they must be mixed well within the ration and sufficient trough space provided, to ensure all animals receive the correct amount. Monitoring is also required to ensure that sufficient acidification is being achieved. Since acidification of the urine occurs within a few days of feeding anionic salts (Husband, 2002), monitoring of urine pH is a routine part of feeding a DCAD diet. This can be time consuming, difficult and in addition to the risk of reduced intakes due to the poor palatability of many anionic salts, as well as the cost, make it a difficult strategy to implement.
The high DCAD value of most pre-calving rations is due to the high potassium content of forages. Even when anionic salts are not fed, measures can still be taken to reduce the DCAD of the diet and thus milk fever risk. When cattle are grazed prior to calving it is important that sward height is kept to a minimum since potassium levels are higher in the tops of the stems than the bottom. In addition pastures that are used to produce dry cow silage or used for grazing dry cows should only be fertilised with straight nitrogen fertiliser rather than slurry, since the latter is potassium rich. In addition later cuts of silage are generally lower in potassium and thus preferential for dry cows.
Oral Supplementation of High Risk Cows:
Even when preventative measures are taken at herd level there is still the potential for cases of milk fever to occur, whether clinical or subclinical. As discussed earlier there are certain cows that are at greater risk than others of disease. As a result, it has become practice by some farmers to routinely give high risk older cows’ intravenous or subcutaneous calcium at calving with a view to reducing the risk of milk fever. However, this is ill-advised since it can be counter-productive as it may slow down her adaptation processes and actually make her more at risk of milk fever due to the negative feedback that a rising blood calcium level has on 1,25-dihydroxyvitamin D3 production.
Where high risk cows are identified then oral supplementation in the form of boluses can be used e.g. Bovikalc. The form of calcium provided in the bolus not only induces a mild metabolic acidosis, thus increasing the resorption of calcium from bone, but also supplies calcium in forms that provide not only a quick release of calcium but also a more sustained slow release so that blood calcium levels are maintained whilst the cow adapts her own metabolism.
Although targeting high risk cows is beneficial, a recent study by Oetzel and Miller (2012) showed that it was not only these cows that benefitted from oral supplementation. In the large-scale study cows from two large, high yielding herds (average 12,400 litres) were enrolled if they were in their second lactation or greater. All cows received anionic salt supplementation during the dry-period and there was historically a low incidence of clinical and subclinical milk fever on the farms (<5%). Cows were divided up into two groups; Control, which received no calcium supplementation at calving, and Treatment, which received one Bovikalc bolus 0-2 hours post-calving and a second 8-35 hours after calving. Cows which were lame, had a low BCS, long dry period or twins/stillbirth were identified as high risk.
Cows were followed through their next lactation to monitor health events, fertility performance and milk yield. The results showed that lame cows and those with higher than average milk yields responded greatest to oral supplementation with Bovikalc. Lame cows in the Treatment group suffered fewer post-parturient health events in the first 30 days in lactation compared to lame cows in the Control group and higher yielding cows also had increased yields at first recording if supplemented. This study should that even when measures are taken at herd level to reduce milk fever risk, such as feeding a DCAD diet, that there are still cows that would benefit from further supplementation.
Summary
Hypocalcaemia continues to be a problem in many herds, not only due to clinical cases but also due to the high prevalence of subclinical cases. Whilst the cost of treating a clinical case may seem low, due to the knock-on effects of low calcium levels on the risk of other post-parturient conditions, prevention is much more cost-effective than cure. There are a number of strategies that can be implemented to reduce the risk of hypocalcaemia at calving, however, there is no one suitable strategy for every farm. Recent research shows that in addition to management at herd level, targeting the individual high risk cow, in particular high yielding and lame cows, for additional supplementation can be beneficial.
This article was kindly sponsored by Boehringer Ingelheim, makers of Bovikalc:



References
DeGaris, P. & Lean, I. (2008) Milk fever in dairy cows: a review of pathophysiology and control principles. Vet J 176(1): 58-69
Grabherr, H. Spolders, M., Furll, M. & Flachowsky, G. (2009) Effect of several doses of zeolite A on feed intake, energy metabolism and on mineral metabolism in dairy cows around calving. J Anim Physiol Anim Nutr 93(2): 221-236
Houe, H., Ostergaard, S., Thilsing-Hansen, T., Jorgensen, R.J., Larsen, T., Sorensen, J.T., Agger, J.F. & Blom, J.Y. (2001) Milk fever and subclinical hypocalcaemia – an evaluation of parameters on incidence risk, diagnosis, risk factors and biological effects as input for a decision support system for disease control. Acta Vet Scanda 42(1): 1-29
Husband, J. (2011) Urine Monitoring – a new tool to prevent milk fever. Farm Business 11th February
Husband, J. (2005) Strategies for the control of milk fever. In Practice 27, 88-92
Oetzel and Miller (2012) J Dairy Sci 95: 7051-7065
Reinhardt, T.A., Lippolis, J.D., McCluskey, B.J., Goff, J.P. & Horst, R.L. (2011) Prevalence of subclinical hypocalcaemia in dairy herds. Vet J 188(1): 122-12
Oetzel, G.R. & Miller, B.E. (2012) Effect of oral calcium bolus supplementation on early-lactation health and milk yield in commercial dairy herds. J Dairy Sci 95(12): 7051-7065
Thilsing-Hansen, T., Jorgensen, R.J. & Ostergaard, S. (2002) Milk fever control principles: A review. Acta Vet Scanda 43:1-19
This article was originally published on VetGrad.co.uk in 2015.

