Show all small animal articles
Long-term management of congestive heart failure due to mitral valve disease
Andrew Francis BVSc, CertVC, MRCVS - 01/06/2013
Long-term management of congestive heart failure due to mitral valve disease
Andrew Francis BVSc, CertVC, MRCVS
Heart disease in dogs is a very common problem in general practice in the UK. According to some previous estimates from the USA, 11 per cent of dogs have heart disease1, and 75 per cent of these cases are thought to be due to mitral valve disease (MVD)2. The incidence of disease increases with age, affecting approximately 20 to 25 per cent of all dogs between the ages of nine and 12 years3.
The definitive underlying cause of the classic valvular contraction, thickening and progressive valvular insufficiency is still unknown, though many theories have been put forward. Genetic factors have been shown to play a large role in the aetiology in cavalier King Charles spaniels and dachshunds4.
An understanding of these mechanisms is key to the development of medications in the future that might treat the actual valve lesions. A detailed discussion of the many theories is beyond the scope of this article, and the answer is likely to be multifactorial.
Although the majority of dogs with MVD never develop clinical signs, about 30 per cent of affected dogs will develop congestive heart failure (CHF)4. Identifying those animals that will deteriorate is very difficult, and those individuals that do develop failure have relatively short survival times of less than one year. In human medicine mitral valve disease is managed surgically, with excellent outcomes for patients5. Unless valve surgery becomes commonplace in veterinary medicine, mitral valve disease in dogs is likely to remain a medically managed disease requiring regular veterinary consultations, treatment with multiple medications, and ultimately clinical deterioration refractory to further medication.
Pathophysiology
Prior to the onset of CHF many dogs will not exhibit any clinical signs easily noticed by the owner6. Initially, cardiac homeostatic mechanisms compensate for the decrease in cardiac output that results from the mitral regurgitation (MR) by increasing heart rate and augmenting cardiac contractility to maintain arterial blood pressure. This results in a decrease in the overall cardiac reserve, necessary during more strenuous exercise, but these dogs remain asymptomatic at rest or during the mild exercise levels common in small breed dogs4. Often, the only indication of a cardiac problem during this phase is the characteristic left apical systolic heart murmur picked up during routine clinical evaluation6.
The mitral regurgitation worsens often over a number of years, and ultimately, systemic compensatory mechanisms become activated via the renin angiotensin aldosterone system (RAAS), though at what point in the disease process this occurs is currently open to significant debate7,8. Circulating blood volume increases, elevating cardiac preload, and systemic vascular resistance is elevated, increasing afterload. Ultimately, heart failure develops once the positive benefit of stable arterial blood pressure is overwhelmed by the unsustainable increased workload on the heart.
Clinical signs
Because a stable arterial blood pressure is maintained throughout the compensated phase of MVD, most dogs present with clinical signs of congestive (backward) heart failure9. CHF can be defined as abnormal fluid accumulation as a result of heart disease9 and common clinical signs include coughing, tachypnoea, dyspnoea, lethargy and anorexia. The severity of the observed clinical signs can be very variable. Some animals present with mild signs and can be managed on an outpatient basis with oral medication; others present with severe, immediately life-threatening clinical signs, requiring hospitalisation and intensive monitoring. Syncope can occur via a number of mechanisms, which are poorly understood9. Sudden death is sometimes seen in cases undergoing treatment for CHF, but is rare in asymptomatic dogs4.
Diagnosis of congestive heart failure
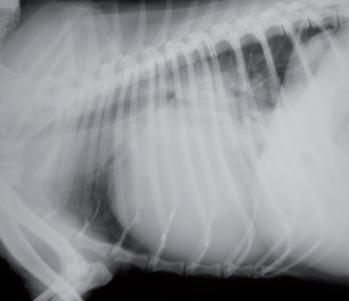
Thoracic radiography is arguably the most important diagnostic tool, provided that the dog is stable enough to tolerate restraint. A perihilar alveolar lung pattern in combination with pulmonary venous congestion, left-sided or generalised cardiomegaly, and radiographic evidence of left atrial enlargement provides a clinical diagnosis of congestive heart failure in most uncomplicated cases (Figures 1 and 2)10. 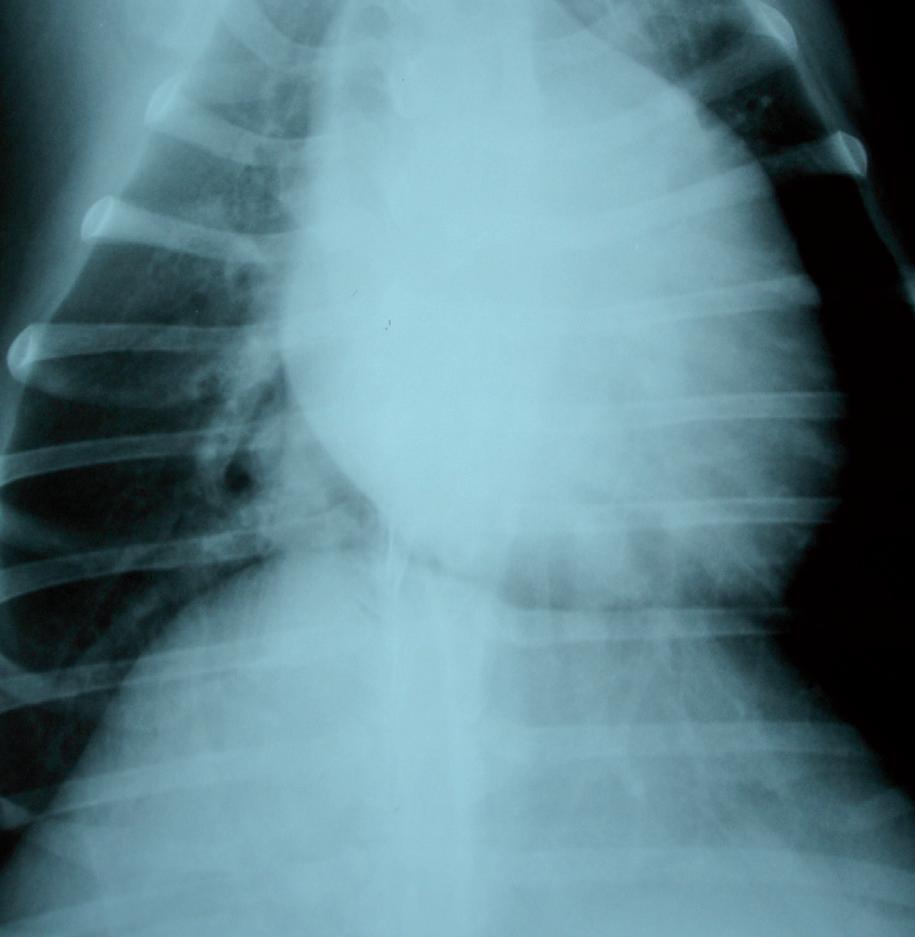
If pursuit of a diagnosis is prevented by the severity of the clinical signs, then oxygen supplementation and a furosemide treatment trial is a viable alternative option.
The identification of mild decompensated heart failure represents a much greater diagnostic challenge because MVD has a long disease course, and animals can decompensate slowly. Most animals will exhibit exercise intolerance on exertion as the disease progresses, but this is often not identified because many affected dogs are old and sometimes obese, and have very little demand on their exercise capacity4.
Cardiac biomarkers are a promising addition to the diagnostic armoury, but they are most useful when asked to answer a specific clinical question, rather than as a general screening test. Canine NT-proBNP especially appears to be a useful marker of the presence of cardiac disease, although values are affected by glomerular filtration rate, and results must be interpreted in the light of the patient’s renal function13.
What treatment do I use?
Ideally, treatment of any disease should follow an evidence-based approach. This is especially important if the condition is immediately life-threatening, because it is essential that medications are rapidly efficacious. More specifically, where possible, selection of specific therapies should be based on the evidence of randomised controlled clinical trials carried out in the relevant patient group9. In veterinary cardiology a significant body of clinical study data has been built up over the past 15 years and was recently reviewed in the ACVIM consensus statement on the guidelines for the diagnosis and treatment of canine chronic valvular heart disease14.
Immediate treatment of animals presenting with acute CHF
- Severe clinical signs.
The mainstay of treatment of acute congestive heart failure is the potent loop diuretic furosemide, though no comparative clinical studies have been performed because its effects are obviously life-saving, and it is considered unethical to withhold furosemide15. It should be given acutely by intravenous injection or constant rate infusion, and titrated to effect in severely affected animals.
Supplemental oxygen is often required in severely affected animals, and judicious use of sedation can be beneficial to improve ventilation if an animal is exhibiting anxiety in association with dyspnoea. Use of Vetmedin® (pimobendan) is recommended by the ACVIM consensus statement at this stage due to strong haemodynamic and experimental evidence14. The suggested dose rate in severely affected animals is 0.4–0.6 mg/kg per day, given in two equally divided doses 12 hours apart and one hour before food. - Milder clinical signs.
Animals with mild clinical signs are often well enough to be treated with oral medication as detailed below and sent home.
Chronic medication
Furosemide should be continued by oral administration at home. The dose rate should reflect the dose of intravenous furosemide used if required for initial stabilisation. Bear in mind that an oral dose of 2mg/kg, for example, would equal a 1mg/kg intravenous dose, due to the 50 per cent bioavailability of oral furosemide.
Vetmedin® (pimobendan) should be used for chronic treatment at home14. At present, pimobendan can be recommended for the treatment of any dog with signs of congestive heart failure secondary to degenerative mitral valve disease16. The QUEST study, still the largest, global double-blinded positive control study in veterinary cardiology, compared outcomes in two groups of dogs with CHF secondary to MVD. The median time to reach the primary endpoint was 91 per cent longer in the pimobendan group† than in the benazepril group†; 267 days versus 140 days17. Evidence from the QUEST study is considered robust enough to alter the priority with which drugs are administered16. Dogs with signs caused by congestive heart failure should always receive a diuretic. In light of the QUEST study, if for any reason only a solitary additional agent is to be chronically administered in addition to diuretics, then Vetmedin® (pimobendan) should be the agent of first choice16. The licensed dose range of Vetmedin® (pimobendan) is 0.2–0.6mg/kg bodyweight per day. The preferable daily dose is 0.5mg/kg bodyweight. The dose should be divided into two administrations (0.25mg/kg bodyweight each), one half of the dose in the morning and the other half approximately 12 hours later. Each dose should be given approximately one hour before feeding. Animals with mild clinical signs can be dosed initially in the range of 0.2–0.4mg/kg per day; this should be increased if no response is seen. In cases with moderate to severe or worsening clinical signs, the 0.4–0.6mg/kg dose range is preferable.
There is currently no published clinical study data that clearly demonstrates the benefit of treatment with furosemide, pimobendan and an ACEi compared with furosemide and pimobendan alone. An appropriately powered prospective clinical study would be required to definitively address this question. Use of all three medications chronically is currently an ACVIM consensus statement recommendation14.
Other medications, including spironolactone, digoxin and other anti-arrhythmics can be added as required.
Progression, clinical monitoring and adjunctive management
Regular clinical monitoring is essential in all dogs with congestive heart failure, both to monitor response to treatment and to document and manage progression of clinical signs. Monitoring can begin at home by teaching owners to monitor their dog’s resting respiratory rate, which is a sensitive indicator of the presence of early pulmonary oedema. A resting respiratory rate persistently greater than 35 to 40 breaths per minute should prompt a consultation for repeat clinical evaluation9.
Many clients will ask for advice on exercise and diet. This can be given on a case-by-case basis, balancing quality of life from routine exercise and a palatable diet, with the changes often recommended, such as mild dietary salt restriction and mild to moderate exercise. Recently, dietary supplementation with omega-3 fatty acids has drawn significant interest in providing support to the heart at various stages19.
Clinical progression can be monitored by serial radiography or echocardiography, though clinical examination, especially evaluation of respiratory rate and heart rate,is a good indicator of clinical progress11.
Clinical deterioration and dose modification
Unfortunately, MVD is an inevitably progressive disease, and gradual clinical deterioration is expected in animals that have previously developed congestive heart failure. In many cases, this deterioration is gradual, but failure to recognise worsening clinical signs can result in a relatively acute, life-threatening decompensation; careful monitoring of resting respiratory rates can allow proactive up-titration of furosemide to help minimise this risk.
Vetmedin® (pimobendan) has a wide licensed dose range, and there is scope for dose modification in the event of deterioration.
Animals previously dosed in the range of 0.2–0.4mg/kg per day should have the dose increased to 0.4–0.6mg/kg per day. The significant survival benefit* achieved in the QUEST study was at an average pimobendan dose rate of 0.5mg/kg per day. The dose rates of other medications should also be re-evaluated whenever there is any clinical evidence of deterioration.
Serum biochemistry to assess affects of medication on renal function should be performed at regular intervals, and within a week of any medication dosage changes.
Refractory congestive heart failure
Ultimately, CHF secondary to MVD becomes refractory to medical management. Clinical signs of CHF persist despite up-titration of furosemide doses, and there is often little sign of active diuresis9. Concomitant clinical signs of right-sided congestive heart failure, such as ascites, are also common in advanced cases9.
Management options in these cases include dose optimisation of currently prescribed medications, removal of any abnormal fluid accumulations that might be adding to morbidity, and addition of other diuretics to target multiple areas of the nephron, known as sequential nephron blockade.
Cardiac cachexia and poor appetite can also be significant problems in as much as half of cases18, and are known to be negative prognostic indicators15.
Currently, the majority of dogs die or are euthanased within a year of the development of clinical signs15. Improvements in survival time in future are likely to be achieved if the time to onset of signs of congestive heart failure can be delayed, though no medications are currently licensed for use in the asymptomatic phase of MVD.
Conclusion
Mitral valve disease is a ubiquitous disease in the canine population. Though many dogs do not develop clinical signs of congestive heart failure, those that do usually live less than one year after clinical signs develop.
The mainstay of clinical diagnosis and monitoring is radiography, though echocardiography and ECG can be very useful in more complex cases, and echocardiography is required for definitive diagnosis.
Chronic treatment with furosemide and Vetmedin® (pimobendan) for congestive heart failure due to MVD has been shown to increase average survival times* by 91 per cent when compared with furosemide and benazepril17. Furthermore, Vetmedin® (pimobendan) is recommended for use in acute, life-threatening CHF14, and the dose can be tailored to the severity of clinical signs because of the wide dose range. Currently therefore, evidence now seems to overwhelmingly support the use of Vetmedin® (pimobendan) in dogs that have developed clinical signs of heart failure secondary to degenerative mitral valve disease, and results from the QUEST study should alter the priority with which drugs are administered16.
†plus furosemide
*time to reach the composite endpoint of cardiac death, euthanasia for cardiac reasons or treatment failure
This article was kindly provided by Boehringer Ingelheim Vetmedica, makers of Vetmedin:

Prescribing information
Presentation: Hard gelatin capsules orange to brown in colour, containing 1.25 mg, 2.5 mg or 5 mg pimobendan per capsule. Presentation: Vetmedin 1.25 mg or 5 mg Flavour Tablets. Meat flavoured tablets containing 1.25 mg or 5 mg pimobendan per tablet. Uses: For the treatment of canine congestive heart failure originating from valvular insufficiency (mitral and/or tricuspid regurgitation) or dilated cardiomyopathy. When used in cases of valvular insufficiency in conjunction with frusemide the product has been shown to improve the quality of life and extend life expectancy in treated dogs. When used in a limited number of cases of dilated cardiomyopathy in conjunction with frusemide, enalapril and digoxin, the product has been shown to improve the quality of life and to extend life expectancy in treated dogs. Contra-indications, warnings, etc: Vetmedin should not be used in cases of hypertrophic cardiomyopathies or clinical conditions where an augmentation of cardiac output is not possible for functional or anatomical reasons (e.g. aortic stenosis).The product should only be used in dogs with cardiac insufficiency. A moderate positive chronotropic effect and vomiting may occur in rare cases. However, these cases are dose-dependent and may be avoided by reducing the dose. In rare cases transient diarrhoea, anorexia or lethargy have been observed. In studies with rats and rabbits, pimobendan had no effect on fertility and embryotoxic effects occurred only at maternotoxic doses. In experiments with rats it has been shown that pimobendan is excreted into milk. No information is available on the safety of Vetmedin in pregnant and lactating bitches. The pimobendan-induced increase in contractility of the heart is attenuated in the presence of the calcium antagonist verapamil and the ß-antagonist propranolol. In pharmacological studies no interaction between the cardiac glycoside ouabain and pimobendan was detected. Do not exceed dosage. In the case of overdose, symptomatic treatment should be initiated. Unused product or waste materials should be disposed of in accordance with current practice for pharmaceutical waste under national waste disposal regulations. For animal treatment only. Legal category: Prescription only medicine. Further information available from Boehringer Ingelheim Vetmedica, Bracknell, Berkshire, RG12 8YS, UK. Tel: +44 (0)1344 746959. Email: vetmedica.uk@boehringer-ingelheim.com
References
1. Buchanan J W. Causes and prevalence of cardiovascular disease. Curr Vet Ther. 1992;XI: 646.
2. Guglielmini C. Cardiovascular diseases in the ageing dog: Diagnostic and therapeutic problems. Veterinary Research Communications. 2003;27 (Suppl. 1): 555–560.
3. Sisson D. Valvular Heart Disease in Dogs. Abstract at WSAVA 2002.
4. Häggström J, Duelund Pederson H and Kvart C. New Insights into Degenerative Mitral Valve Disease in Dogs. Veterinary Clinics of North America: Small Animal Practice Volume 34 (2004) Pages 1209-1226.
5. Detaint D, Sundt T M, Nkomo V T, et al. Surgical Correction of Mitral Regurgitation in the Elderly: Outcomes and Recent Improvements. Circulation 114 (4), 2006 Pages 265–272.
6. Kittleson MD and Kienle R D. (1998) Myxomatous Atrioventricular Valvular Degeneration. In: Small Animal Cardiovascular Medicine pp 297–318. Mosby Inc, St Louis.
7. Häggström J, Hansson K, Kvart C et al. Effects of naturally acquired decompensated mitral valve regurgitation on the renin-angiotension-aldosterone system and atrial natriuretic peptide concentration in dogs. American Journal of Veterinary Research 1997; 58: 77–82.
8. Oyama M and Sisson D. Blood based detection of occult heart disease. In: Proceedings of the 21st Annual American College of Veterinary Internal Medicine Forum. Charlotte, NC, June 4th–8th, 2003. p. 88– 89.
9. Luis Fuentes (2010) Treatment of congestive heart failure. In: BSAVA Manual of Canine and Feline Cardiorespiratory Medicine 2nd edn, ed. Luis Fuentes V, Johnson L R, Dennis S, pp 153–159. British Small Animal Veterinary Association, Gloucester.
10. Häggström J (2010) Myxomatous mitral valve disease. In: BSAVA Manual of Canine and Feline Cardiorespiratory Medicine 2nd edn, ed. Luis Fuentes V, Johnson L R, Dennis S, pp 186–194. British Small Animal Veterinary Association, Gloucester.
11. Borgarelli M, Häggström J. Canine Degenerative Myxomatous Mitral Valve Disease: Natural History, Clinical Presentation and Therapy. Topics in Cardiology Veterinary Clinics of North America: Small Animal Practice Volume 40, Issue 4, July 2010, Pages 651-663.
12. Kittleson MD and Kienle RD. (1998) The approach to the patient with cardiac disease. In: Small Animal Cardiovascular Medicine pp 195–217. Mosby Inc, St Louis.
13. Boswood A, Dukes McEwan J, Loureiro J. et al. The diagnostic accuracy of different natriuretic peptides in the investigation of canine cardiac disease. Journal of Small Animal Practice (2008) 49 pp 26–32.
14. Atkins C, Bonagura J, Ettinger S et al. Guidelines for the diagnosis and treatment of canine chronic valvular heart disease. ACVIM Consensus Statement. Journal of Veterinary Internal Medicine (2009) 23 pp 1142–1150.
15. Häggström J, Hoglund K and Borgarelli M. An update on treatment and prognostic indicators in canine myxomatous mitral valve disease. Journal of Small Animal Practice (2009) 50 (Suppl. 1). pp 25–33.
16. Boswood A. Current use of pimobendan in canine patients with heart disease. Topics in Cardiology Veterinary Clinics of North America: Small Animal Practice Volume 40, Issue 4, July 2010, Pages 571–580.
17. Häggström J, Boswood A, O’Grady O, et al. Effects of Pimobendan or Benazepril Hydrochloride on Survival Times in Dogs with Congestive Heart Failure Caused by Naturally Occurring Myxomatous Mitral Valve Disease: The QUEST Study. Journal of Veterinary Internal Medicine (2008) 22: 1124–1135.
18. Freeman L M, Rush J E, Kehayias J J, et al. Nutritional alterations and the effect of fish oil supplementation in dogs with heart failure. Journal of Veterinary Internal Medicine (1998) 12 440–448.
19. Freeman L M. Beneficial effects of omega-3 fatty acids in cardiovascular disease. Journal of Small Animal Practice (2010) 51, 462–470.

