Show all small animal articles
Conservative Management of Canine Osteoarthritis
Samantha Lindley BVSc MRCVS - 14/12/2010
CONSERVATIVE MANAGEMENT OF CANINE OSTEOARTHRITIS
INTRODUCTION
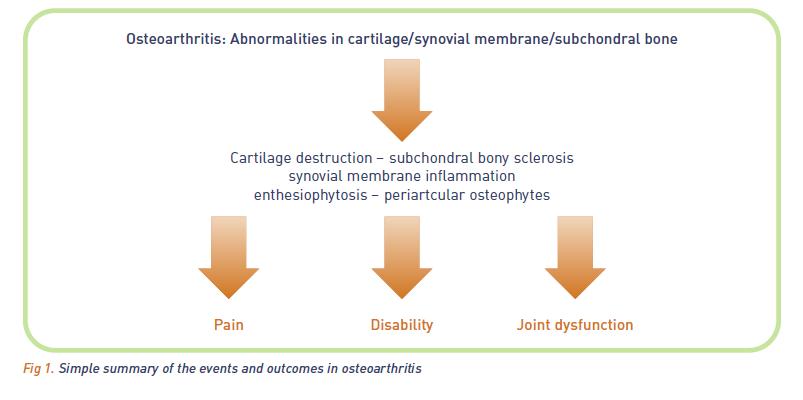
Osteoarthritis (OA) is often defined as “a progressive, degenerative disease of synovial joints characterised by pain, disability, destruction of articular cartilage, and bony remodelling”. However, this relatively straightforward definition can often be misleading, especially when the complex biochemical, pathological and physical changes caused by OA are taken into consideration. This complexity confounds our ability to find a simple, straightforward solution to canine OA, which often frustrates clinicians and dog owners alike. Dog owners understand that canine OA can be debilitating, adversely affecting both the dog’s and the owner’s lives. However, canine OA does not always have to cause significant suffering, nor be the grim predictor of a shortened lifespan. This article details canine OA and its diagnosis, the assessment of chronic pain, and the wide range of available therapies.
 OA IN THE DOG – IMPLICATIONS FOR PATIENT, OWNER AND PRACTICE
OA IN THE DOG – IMPLICATIONS FOR PATIENT, OWNER AND PRACTICE
Canine OA is usually a secondary condition (i.e. secondary to conformational abnormalities such as hip dysplasia, elbow dysplasia, and osteochondrosis), differing from human OA and feline OA, which are usually primary (i.e. caused by ‘wear and tear’). What this means for the canine patient is that clinical signs and radiographic changes may appear when the dog is still very young (Figure 2), potentially resulting in the following implications:
• Pain at a young age
• Impaired exercise tolerance leading to poor development of muscle and gait
• Untreated, or undertreated, pain at any age is a chronic stressor (see below) which affects posture, behaviour and may lead to a chronic pain state
• Pain during the behavioural development phase may manifest as aggression or anxiety, avoidance behaviour and poor or inadequate socialisation
• Postural and gait abnormalities put strain on other musculoskeletal structures and predispose to pain, possibly accelerating the development of primary or secondary osteoarthritis in other joints
• Poor exercise levels will predispose to obesity, exacerbating pain
For the owner, the implications of early development and/or diagnosis of OA are:
• Shock and distress that such a young animal develops a condition that they usually associate with old age and debility
• Negative comparisons between their pet’s condition and other arthritic conditions with which they are familiar in humans (e.g. rheumatoid arthritis)
• The assumption that this diagnosis represents a death sentence, or at least a shortening and reduction in quality of their dog’s life
• Disappointment, as they cannot share the experience of their dog exercising and playing normally
• Worry that their dog is suffering
• Worry about the potential side effects of medication
• Worry that the medication will inevitably be life-long and may shorten life
• The sense that they have no control over the problem
For the veterinary practice, the implications of the diagnosis of OA in a young dog are:
• That the owner must be reassured and quickly given the tools with which to cope with their pet’s condition
• Involvement, for the life of the patient, in the dynamic process of managing OA
• Accepting that this condition cannot be cured or “fixed” and that chronic conditions may easily become the classic “heart sink” cases
• That the “heart sink” scenario should not be allowed to develop by always having access to resources that can add to the management plan, by frequent reassessment of the patient’s condition, by improving pain relief and by giving owners a sense of control over the problem
• That the whole team can, and arguably should, be involved in the process
DIAGNOSIS OF OA
PRESENTATION
Patients may be presented to the practice with a variety of clinical signs and at any age:
• Lameness
• Stiffness
• Limping
• Gait abnormalities (e.g. “bunny hopping” or “crabbing”)
• Scuffing of the nails (forelimb or hindlimb; Figure 3)
• Excessive licking of a limb
• Swelling around a joint (Figure 4)
• Inability to do one or more certain movements (e.g. jumping, going up and downstairs, getting in and out of the car, getting up, lying down, sitting down)
• Change in exercise tolerance
• Changes in behaviour (see below)
N.B. Behavioural changes/problems may be the only sign of pain and disease in an ostensibly healthy animal
CLINICAL EXAMINATION
In addition to the standard clinical examination for general health, the whole dog should be examined, not just the limb that appears to be giving the problem. An initial scan with light touch may pick up any gross abnormalities, such as masses or gross differences in muscle atrophy and swollen or deformed joints. Just as importantly, this initial scan will help to pick up areas of allodynia (response to light touch as though it were a painful stimulus). This would be indicative of the animal suffering from a chronic pain state (see below), which would be missed if the limbs were grasped firmly on initial examination. Allodynia can be identified by a shuddering or fasciculation of the affected area in response to light touch and/or by a flinching away from the touch; in severe conditions, and depending on the individual, such touch may also elicit aggression.
As a general rule, examination should start at the furthest point from the area that is suspected to be the most painful. As an additional guide, if the left elbow, for example, is the joint suspected to be affected by OA, then start at the ipsilateral hindlimb, because the weight will usually be thrown back onto the contralateral hindlimb, as well as onto the opposite forelimb. The rationale behind this approach is twofold:
1. If the most painful area is examined first and the patient is hurt then, effectively, the examination is over, because no further useful information will be gathered; muscles will be tense, joint movement will be restricted and the patient may react at a lower threshold than previously.
2. This examination may reveal that the main source of pain is in fact in a different place than first suggested by the initial presentation. For example, forelimb lameness may be the initial presenting sign, but examination may suggest that the hindlimbs, or the lumbar area, is significantly more painful.
Forelimb lameness (except for traumatic injuries) should always prompt the clinician to look at the hindquarters. Given that the dog carries approximately 75% of its weight on its forequarters, hindquarter pain will significantly increase that burden. Weight thrown off the hindlimbs first puts strain on the lumbar muscles and then the forelimbs, causing muscle strain and presumably exacerbating the effects of any pre-existing forelimb pathology. This phenomenon should also be suggested if the dog is having more trouble going upstairs than down, into the car rather than out, and uphill rather than downhill, and/or rises by first sitting up and then obviously hauling its hindquarters off the ground with its forequarters.
The examination will then continue by the examination of the joints, examining for:
• Swelling (bony or fluid)
• Bony distortion
• Heat
• Crepitus
• Pain on direct palpation and on palpation above and below the joint
N.B. Not all joints with arthritis and not all joints with painful arthritis will show these changes (i.e. the absence of these signs does not rule out arthritis)
RANGE OF MOVEMENT
This can be difficult to assess and tends to improve on repetition unless the dog finds it painful in which case it may reduce with subsequent attempts. A goniometer will accurately measure range of movement and “normal” ranges of movements for the joints are available.
Bear in mind that a reduced range of movement does not necessarily equate to a painful joint. In particular, most dogs do not take every joint to the extreme of flexion and extension during normal movement. Full extension of the arthritic/dysplastic hip may elicit a painful response, but does not mean that the dog is inevitably suffering that degree of pain except when that particular movement is carried out. Repeated “testing” of the efficacy of any analgesia by performing this extension on every examination is not only unhelpful but also unnecessary and unkind.
MUSCLE ATROPHY
Muscles that are not being used properly will atrophy. A significant clue to the restriction of joint movement by pain or mechanical limitation will be the amount of muscle atrophy that is present in the relevant muscle groups. This is harder to assess in bilateral lameness and in dogs with cachexia (neoplastic/cardiac) or concurrent disease, such as protein-losing nephropathy/enteropathy/hepatopathy. Swapping hands so that the same muscle mass is assessed by both hands (not just the dominant one) makes the assessment clearer since the dominant hand tends to be less sensitive.
MUSCLE PAIN
The detailed assessment of muscle pain is not usually taught at veterinary or medical schools. Myofascial pain is a complex area that is outside the remit of this article, but many references are available.1,2 Myofascial trigger points (MFTrPs) are areas in muscle that are hyperirritable loci within taut bands or “knots” in muscle.
These bands are painful on compression, can elicit a jump sign (involuntary movement away from the pain), a twitch response in the local muscle and a characteristic referral pattern of pain (described in humans) when compressed. Essentially, they equate to the “knots” in muscles that masseurs and physiotherapists may find when examining muscle.
Trigger points arise in response to muscle strain, postural strain, radiculopathy and other sources of somatic and visceral pain. A painful arthritic joint will give rise to painful MFTrPs in the muscles local to the joint. The postural shift which occurs when an animal takes the weight off a limb will also cause trigger points in other muscles (the ones which are now taking the strain), but one would not expect these to be as painful on palpation as those associated with a painful joint. Trigger points appear to arise predictably in certain muscle groups (e.g. painful elbow arthritis will be associated with painful trigger points in triceps, infraspinatus and caudal trapezius muscles; painful hip arthritis will be associated with painful trigger points in gluteals, rectus femoris and longissimus muscles).
Muscle pain can be widespread and debilitating and will contribute to the suffering caused by OA. The more intense the pain and the longer the duration, the more widespread is the pain referral from muscle.3 Note that MFTrPs are not inflammatory and neither do they equate to “muscle spasm”. They sometimes appear to be refractory to many analgesics, although if the original source of pain is treated they may “settle down” to become “latent” rather than “active” trigger points. Palpation of these trigger points is a practical skill that needs to be demonstrated rather than described.4
CONFIRMATION OF OA DIAGNOSIS
Confirmation of OA is by radiography. In the dog the radiographic picture is usually associated with an osteophytic reaction (unlike in cats, where osteophyte production is modest by comparison).
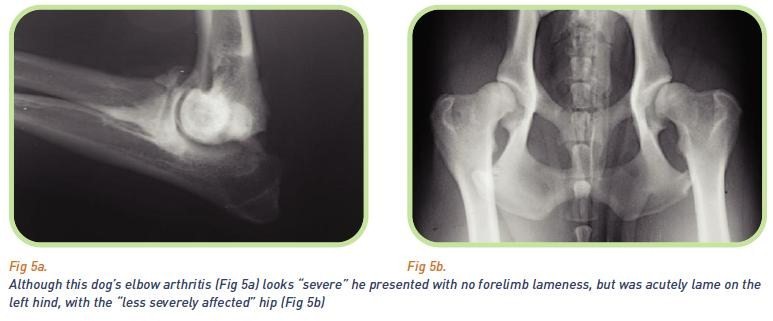
N.B. A confirmation of OA is NOT a confirmation of pain and suffering (although the animal should be given the benefit of the doubt until proved otherwise). Furthermore, the severity of radiographic changes in no way predicts the degree of pain and suffering caused by the arthritis.
If untreated, OA is a problem to the patient in two main areas:
1. It is often a painful condition and this affects quality of life by causing suffering.
2. The function of the joint becomes increasingly impaired to the extent that it eventually becomes nonfunctional. This has an impact on the local muscles, other areas of the body (as discussed above) and in other less obvious ways, such as abnormal wearing of nails and excoriation of the skin on the dorsum of the paw. These may, in some cases, cause the animal more immediate suffering than OA itself.
Veterinary intervention potentially creates problems for the patient and owner in other areas:
1. Restriction of the dog’s resources by limiting exercise and the kinds of play in which they can engage, potentially causing frustration and confusion. This restriction can also cause problems for the owner because they feel guilty about restricting an activity from which their dog derives pleasure.
2. Food is a major resource that is often restricted or changed. Owners will often feel guilty about limiting their animal’s food if they “look hungry” and are dissatisfied if they do not enjoy their food. The relationship between the provision of food and affection is a complex one and such feelings often negatively affect owner compliance.
3. Potential side effects of medication may affect the wellbeing of the patient, yet, whether the side effects occur or not, this may worry the owners.
These potential pitfalls are covered later in this article under therapeutic approaches. This section is to remind the reader of the challenges of pain in OA.
ACUTE PAIN
The experience of acute pain is vital to survival; an organism must experience the sensation we call pain to withdraw from the painful stimulus (and therefore limit the damage), protect the damaged area and learn not to repeat the experience, if possible.
However, this is not a reason to leave patients in acute pain or to use pain to limit movement and activity.
Ignoring, or under-treating, acute pain not only causes immediate suffering but is likely to lead to prolonged, chronic pain and the development of chronic pain states. It is unlikely that any analgesic will provide powerful enough analgesia to allow an animal to be able to exercise normally in the face of significant pathology.
CHRONIC PAIN
The experience of pain is not a straightforward transmission-perception process from the peripheral stimulus to the brain. This is true of acute pain, where there are many levels of modulation of pain from the periphery, through spinal cord to brain. The experience of acute pain from the same stimulus will be experienced differently by different individuals.
In chronic pain the picture is even less clear:
Chronic pain is not adaptive, i.e. it is of no biological use to the animal. This is important to recognise, because owners often still think that pain is somehow of use to the patient and that they do not want to be giving something that “just masks the pain”.
Chronic pain causes the same physiological changes as chronic stress: poor quality sleep (which leads to more pain), increased heart and respiratory rates, increased blood viscosity, muscle fasciculation and trembling.5 Untreated pain therefore exposes the patient to the physical and psychological effects of ongoing chronic stress.
In chronic pain, because of the “neuronal plasticity” of the nervous system (i.e. the way that pain is perceived is not “hard wired”) the pain is not only part of the disease but can become the disease. The experience of pain may continue to get worse for the patient even if there is no change in the pathology, or sometimes even when the pathology is removed (phantom limb pain is the clearest example of this). This is due to the changes in the nervous system where increasing numbers of systems become involved in pain signaling (wide dynamic range neurons (WDR), NMDA receptors and glutamate receptors, causing the up-regulation of further receptors, membrane depolarisation and the experience of further pain). The normal physiological consequence of this is spinal “wind up” which occurs in every mammal and may last minutes or hours depending on the cause and the current state of the nervous system. If this process continues or goes on unchecked then central sensitisation may result; here there are transcriptional changes in dorsal horn neurons leading to altered neurotransmitter levels at synapses and an increased number of receptors. This state may include an imbalance between excitatory and inhibitory influences (central disinhibition) and changes to the A-beta fibres that usually “recognise” light touch but now produce substance P, which causes input from these receptors to be perceived as pain.6 It is not fully understood why this happens in some individuals and not in others, but at least one risk factor is likely to be uncontrolled pain. The extent of suffering in these patients is completely uncorrelated to anything one might see on imaging and may exist without significant pathology.
In OA, the dynamic nature of the condition, and the difficulty of recognising pain in patients who cannot verbally complain about it, means that it is likely that dogs will experience uncontrolled pain from time to time, even if some analgesia is given. Under-dosing, using analgesics only when the patient appears to be really suffering (what the owner tends to do), or just giving short, limited courses of analgesics, is likely to predispose at least some of these dogs to a chronic pain state or central sensitisation. Repeated acute “flare ups” may well contribute to this problem and this is one of the best reasons to avoid a “fire fighting” approach to the treatment of OA.
SOURCES OF PAIN IN CANINE OA
THE JOINT
In soft tissues of the joint, the synovial membrane becomes inflamed (“synovitis”), leucocytes enter the joint space and enzymes, cytokines, free radicals and prostaglandins are released, triggering further inflammation. This inflammatory process damages the cartilage.
The cartilage erodes due to damage to the supportive matrix and further inflammation. This process means that it is less able to resist mechanical injury and the subchondral bone is eventually exposed. Whilst cartilage has no nerve supply, bone is richly innervated and the pain is further increased when it is exposed.
THE SOFT TISSUES
Secondary muscle pain mainly consists of myofascial pain in local muscles and those that take the strain of the postural shift off the affected joint or limb. Note that these may continue to cause pain for the patient even when the joint is not in a chronically active inflammatory state.
CENTRAL CHANGES
These include central sensitisation/disinhibition as outlined above. Note that these may continue to cause pain for the patient even when the joint is not in a chronically active inflammatory state.
N.B. Available evidence suggests that a reduction in central sensitisation can reduce peripheral disease and inflammation (i.e. treating pain effectively may also reduce inflammation and disease progression).7
APPROACH TO TREATMENT
The treatment of OA has two main aims:
1. To drive the chronically active arthritic joint into a chronically silent state (i.e. where there is minimal inflammation and pain)
2. To slow, as far as possible, the inevitable progress of the joint to its dysfunctional end stage
Traditionally, the approach to OA relies on diagnosis, assumed or confirmed, followed by...
...ANALGESICS, USUALLY NSAIDs
These are prescribed either short-term to cover acute flare-ups, when the condition is first diagnosed, or, in contrast, permanently. The problem with the short-term approach is discussed above; it is likely that the patients are not receiving sufficient analgesia and are being predisposed to chronic pain states because they are exposed to repeated bouts of pain. The problem with permanent therapy is that owners may perceive this to be “over treatment” and then either only resuming therapy (which then somtimes appears not to ‘work’) when the pain has become as severe as before, or revisiting the practice. There is an instinct that warns the owner against long-term medication, especially in a young dog, and this practice must be rationalised and justified to the owner.
This can be achieved by:
a. Assessing the extent of patient’s suffering (see below), so that the owner understands why medication is being used (or not used).
b. Recording realistic outcome measures with the owner’s help, so that the owner can look for specific improvements or deteriorations and understand why medication may be maintained or altered.
c. Tailoring the dosage and frequency of treatments according to individual response.
This way the owner becomes part of the treatment programme and sees the reasoning behind why their dog may sometimes be given long-term therapy, as well as understanding why that treatment is sometimes suspended or increased.
...GLUCOSAMINE/CHONDROITIN
After the initial course of NSAIDs it is often hoped that this nutraceutical combination may continue to manage the patient’s problem and limit the progress of arthritis. The evidence for either of these being achieved with glucosamine/chondroitin as a sole treatment is, at best, currently equivocal (in both dogs and humans).
Since safety is not a factor in treatment with these veterinary formulated compounds, there is no reason to stop using them as part of the management of OA, but, on the strength of current evidence, it would be hard to justify them as a sole medication if the animal is suffering OA pain.8,9
...EXERCISE
Limited exercise is the usual advice, with regular, short walks and no weekend hikes. This is correct at the time of initial diagnosis and if the dog is suffering. However, in the absence of pain, many dogs with OA manage to increase their exercise over time to normal levels. This helps to regulate weight, improve muscle tone, bulk and strength and generally improve wellbeing, as well as being pleasing for the owner. Advice about exercise should therefore not stop at “restricted” but, again, be tailored and adapted to the level with which the dog can cope.
But, the management of OA can offer so much more than this to the patient and the owner. The key is to appreciate the dynamic nature of the condition and therefore its treatment.
A NEW APPROACH TO MANAGING CANINE OA
ASSESSMENT... not just of pain but also of suffering
The individual tailoring of a programme relies in part on being able to assess the patient’s suffering. This assessment guides the clinician in deciding how much should be done and how quickly, and can help balance questions of quantity versus quality of life.
All of the validated pain scores rely, to a greater or lesser extent, on the assessment of behavioural changes in response to pain and on more than one assessment. The Glasgow Composite pain scale uses a number of outcome measures and has an intervention score at which additional pain relief should be considered.10 This scale is currently validated for acute pain in orthopaedic patients. A quality of life scale, validated in orthopaedic patients with chronic pain, is currently being revised and shortened.11 This subject has been covered in detail recently by Yeates and Main.12
In this author’s pain clinic the technique dubbed “triangulation” is used to assess the degree of suffering in the pain clinic patient. To be accurate, pain and suffering are not synonymous – pain is the sensation, but suffering is the negative way it causes the animal to feel about the sensation. For example, a dog with an arthritic stifle may have a sensation of pain in that joint when he runs or when he walks upstairs, or when the veterinary surgeon palpates or moves it, but that does not stop him performing those activities, nor does it cause him to have aversive feelings about that sensation, thereby learning to avoid certain behaviours that cause it. Neither does that sensation keep him awake, or stop him lying comfortably, or make him anxious about other dogs coming towards him. Therefore, there is pain but no significant suffering. However, the dog that has the sensation of pain plus all the aversive and unpleasant associations that go with pain, is suffering. The first example needs relatively simple pain relief, such as NSAIDs, to allow as normal movement as possible and to stop the pain progressing toward suffering. The second example needs aggressive multi-modal analgesia, attention to its comfort and appreciation of the fact that some normal experiences (such as grooming or interacting with other dogs) are unpleasant and even frightening.
This is the key to approaching the pain of OA; the assessment of suffering. Once there is an appreciation of how much suffering is present, the risk-benefit analysis of using multiple, and sometimes off-licence, medications is simplified, even in the presence of concurrent disease. Few owners would want their dog suffering as much as the example above, even if treatment of that suffering created some measure of risk to organ function, but if the first dog were in an advanced stage of renal failure and being managed well, one would feel justified in carefully exploring methods of pain relief that were unlikely to further compromise renal function.
TRIANGULATION
This technique starts from the premise that one can never be certain about an absolute value of the degree of suffering in an animal (or even in a human, despite our verbal dexterity), just as one can never give a certain value on an animal’s welfare. It is what John Webster, in his book “Limping Towards Eden”,13 describes as a ‘fuzzy area of science’. What is needed in such areas is a way of decreasing the uncertainty around one’s assessment. Webster uses a sailing analogy to illustrate the point: if one is trying to work out where one is whilst at sea, there is little use in taking only one bearing (or using a measure of cortisol as a single outcome measure of “stress”) because there still remains too much uncertainty. Two bearings are better, but three further reduce the area of uncertainty about the position. The composite pain scale described earlier uses a similar approach – by measuring different expressions of animal pain individual variation is minimised; if touching a wound is the only measure of pain then what about those animals who are frightened or who dislike being touched by strangers anyway?
The Pain Clinic uses triangulation in the following way:
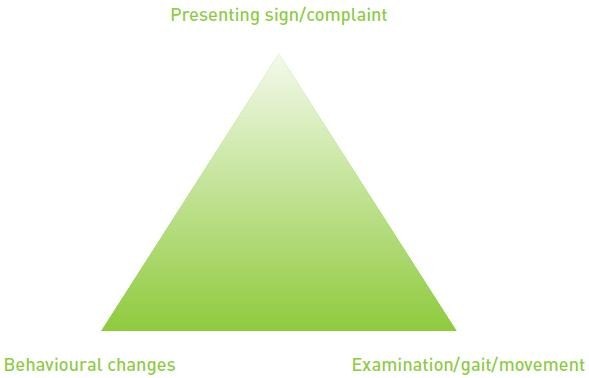
the presenting complaint or sign is the starting point for most consultations
In OA, for example, the owner may present a dog that can no longer jump into the car, or for whom on survey radiographs of the pelvic area OA of the hips is identified. The dog may not want to jump into the car for a variety of reasons: it may feel car sick, it may have had a bad experience when being left in the car, the car may have been changed and now simply be much higher or more awkward to get into, the dog may have learned that reluctance to get in the car receives encouragement in the shape of a food reward, or its sibling dog may be quietly, but firmly, discouraging its entry. Similarly, the identification of arthritic changes in the hips is suggestive of pain, but does not absolutely mean that there is pain – or suffering – present. Therefore, taking this single presenting sign or complaint as an indication of pain and suffering could result in over, or inappropriate, treatment for pain.
The second bearing is that of behavioural changes for that individual. Are there changes other than reluctance towards getting in the car? A list of possible changes is given in Table 1. However, it should be borne in mind that such changes alone do not indicate pain. Spending more time with the owner, or being “clingy”, may occur in response to an environmental change such as moving house, or a family member moving out, or a sibling dog dying. Such changes always need to be checked against possible environmental or social causes, e.g. trying to hide from other dogs may be the simple response to a recent dog attack.
exercise tolerance
enthusiasm to start exercise
willingness to play with owner
willingness to play with sibling dog
change in who initiates play
more clingy or more aloof
changes in aggression
changes in anxiety
change in sleeping patterns
restless when trying to sleep or settle
pacing and/or panting
jumping/starting as though stung or startled
frequently looking round at a body area
frequent nibbling or licking at a body area
change in attitude to grooming
change in attitude to travel in the car
change in ability to be left alone
change in greeting the owner in the morning or when they return
change in appetite
change in sound sensitivity
change in attitude to young children
change in attitude to young or boisterous dogs
change in attitude to dogs generally
change in attitude to people generally
N.B. Other environmental or social causes may also result in such changes and must be checked
Table 1. Behavioural changes likely to arise as a result of pain in dogs
The third bearing is taken from both the examination and the gait and/or general movement. Although these could strictly be two separate bearings, the pain clinic patients are not restricted to patients with musculoskeletal pain, and non-ambulatory patients also need to be assessed for pain and suffering. The examination process has been described earlier in this article. Gait and movement should be assessed outside, if possible: on slopes, up and down steps and at different speeds. This assessment can be limited by patient cooperation and the gait of a dog desperately pulling to get away from the clinic is not the easiest to judge. If this is a limitation, then asking the owners to video the dog at a walk, trot and run, up and downstairs etc. can be helpful, particularly if other parts of the overall assessment are not straightforward. Slowing down the video can also make it clearer to determine on which leg the patient is lame.
As well as frank lameness, stiffness, pacing, inability to pick the feet up properly, a shortened stride and difficulty in turning tight circles can also be picked up at this time and may indicate additional problems or the need for a neurological examination. This part of the assessment is a useful point at which to ensure that the owner and the assessor are in agreement about the site of lameness and getting the owner to point at which leg they think is the problem can offset a raft of potential confusion and miscommunication.
TREATMENT
Treatment of OA should incorporate not only treatment of pain, but an attempt to limit dysfunction for as long as possible, to limit the frustration of the patient from having its resources restricted, and to limit the guilt felt by the owner from having to limit these resources and administer medication about which they feel ambivalent.
Above all the approach must be dynamic and, where possible, proactive.
Carmichael describes an approach that fits these requirements.14 The straightforward A, B, C, D, E mnemonic provides an instant checklist for the practitioner in the treatment of OA:
A = Analgesia
B = Bodyweight
C = Control (i.e. Control of complications and of comfort, and giving the owner a sense of control. It also stands for common sense. To paraphrase Carmichael;14 “If the dog is running about freely and happily and the only thing it cannot do is get in the car, then the dog needs a ramp and not a hip replacement”.)
D = Disease modification
E = Exercise
Carmichael and Lindley have consolidated this approach in the Pet Arthritis Counselor Training course:
A = Analgesia
NSAIDs are the first line of approach for dogs with OA. NSAIDs, as it is well known, act partly by decreasing prostaglandin synthesis by inhibiting the enzyme cyclooxygenase (COX) which occurs in two forms: COX 1, the so called constitutive COX, and COX 2 which is induced during cell injury and inflammation. The general trend in veterinary pharmaceuticals is towards preferential COX 2 inhibition since, although the story is not so clear cut as to label COX 1 “all good” and COX 2 “all bad”, there is increasing evidence that significantly more COX 2 has been found in the synovium of OA affected hips than normal joints in dogs.15,16 COX 2 is also implicated in the development of central sensitisation and inhibition of COX 2 has been shown to decrease central sensitisation.16
The administration of a COX 2 inhibiting NSAID is now taking on a further potential importance for the patient: it should specifically target the inflammatory process in affected joints and it should decrease central sensitisation, therefore having an impact on chronic pain states. From the evidence that if central sensitisation is decreased, peripheral disease and inflammation are reduced,7 it may be hypothesized that COX 2 inhibiting NSAIDs may actually help to treat the disease by treating the pain.
Each NSAID has a different profile, differing pharmacokinetics and, therefore, it is not surprising that on occasion, different individuals respond differently to a variety of NSAIDs. Sometimes this varying response is in terms of side effects, and sometimes in terms of apparent efficacy. Some individuals appear to respond predictably and adversely to all NSAIDs, and some only to one. It is claimed that some patients no longer have a benefit from a given NSAID after a while (so called “tolerance”), and a change to another NSAID may elicit an improved response. It is possible that the patient may always have been likely to respond better to the alternative drug and that its condition was simply getting worse, such that the first no longer provided sufficient analgesia. Without reliable and consistent pain scoring, it would be impossible to say whether this effect is real or just due to the vagaries of chronic pain, individual response to NSAIDs, and/or the progression of OA. There appears to be no logical mechanism by which such “tolerance” of these drugs occurs. It is also not infrequent that the owner realizes that the first NSAID was actually having an effect when the replacement NSAID appears to have no effect at all on their animal’s pain.
Side effects of NSAIDs are well documented, and include gastric irritation and ulceration, kidney failure and hepatotoxicity. Whilst caution is sensible in the face of any gastrointestinal disturbance if its origin is not known, it is probably true to say that NSAIDs are withdrawn too often in response to vomiting and diarrhoea. Unless there are signs of gastric ulceration (which would obviously indicate treatment in its own right), it is probably wise to withdraw the NSAID whilst the problem is present, and then reintroduce it when the signs are resolved.
One side effect from NSAIDs that is not much discussed is nausea. It is not known whether there is a connection between nausea and gastric ulceration caused by NSAIDs, or whether nausea is a risk factor for ulceration. However, nausea itself is a highly unpleasant experience, and replacing pain with nausea is not likely to improve the animal’s wellbeing. Signs of nausea include an increase in grass or plant eating (pica is also possible), an increase in lip licking, excessive yawning and stretching (although the later may also indicate abdominal pain/low back pain), inappetence and general dullness of demeanour, with or without vomiting. These signs should be checked for by questioning the owner at each consultation.
MULTIMODAL ANALGESIA
Pain is modulated at various sites in the nervous system and, at its simplest, multimodal analgesia exploits the different actions of different analgesics to maximise the effect on pain, and minimise the overall amount of each analgesic. Whether an animal needs analgesia additional to NSAIDs will depend on the assessment of its suffering. It is not a given that all animals need multiple analgesics nor that all patients with OA will need analgesics all of the time, hence the need for frequent re-evaluation and adaptation of the individual approach.
There are many unlicensed analgesics in common use for pain in small animals. On one hand, it is reassuring to know that there is recognition that NSAIDs are not always enough and that many animals may be suffering, on the other, there is a risk that the norm simply shifts to a new protocol. Where it used to be long-term NSAIDs and glucosamine/chondroitin (or prednoleucotropin, or glucocorticoids, where NSAIDs did not work) the new “formula” may become NSAIDs, plus tramadol plus gabapentin.
Although there is no proof of the efficacy of these for the treatment of pain and suffering in dogs with OA, the impression is that they have had a significant effect on the welfare of patients suffering from pain arising from a variety of sources. However, that does not mean that no circumspection is required in their use; excessive use of these medications will result in owners perceiving that their pets are being “doped up” and, of course, the more sedating drugs used, the more likely it will be that an individual will be sedated. Dullness and sedation will complicate continued assessment of the pain and suffering.
Analgesics employed in multimodal analgesia...
- Tramadol hydrochloride... this is often thought of as an opiate, but has only weak opiate activity, binding at μ-receptors, and also interacting with both noradrenergic and serotonergic systems, as well as having other central effects. Serotonin and noradrenaline are neurotransmitters involved in two of the descending inhibitory pain pathways that are essential in controlling and inhibiting the response to painful stimuli. It is important to remember that tramadol is not classed as an anti-inflammatory and, if one suspects that active inflammation is present, then an anti-inflammatory should be used, except when contraindicated. However, there is some evidence that central inhibition of pro-inflammatory cytokines may occur with the use of tramadol, and, with increasing evidence that the inhibition of central sensitisation leads to a reduction in peripheral disease, this view may change. Tramadol is best used where the patient is suffering (this distinction is made earlier in the article), in short, where the pain affects its mood and demeanour. Although tramadol may have an effect on lameness, it is not unusual to see that the dog just limps more happily i.e. one may use a different outcome measure when treating with tramadol.
Since the drug is not licensed for use in animals, a comprehensive list of side effects and contraindications is not available. Side effects seen include sedation, dullness, vomiting, nausea, diarrhoea and, specifically, dysphoria. It is associated with lowering seizure threshold in man, but this does not appear to be a problem in dogs, whereas seizures have been reported in cats. The dose commonly used is 2-5mg/kg bid-tid, although starting at the lower dose range often has an effect and will limit side effects. The drug appears to be highly stable and can be used in its powder form or dispensed into gel capsules for dosing smaller dogs. Additionally, it appears to be safe to use with NSAIDs. It would be wise to keep the doses to a minimum in either renal or hepatic dysfunction. Tramadol should not be used with other serotonergic drugs such as clomipramine, amitryptline or monoamine oxidase inhibitors, because of the potential danger of causing serotonin syndrome. - Gabapentin... this is classed as an anti-convulsant and was first licensed for neuropathic pain in humans. Neuropathic pain involves the nervous tissue directly, such as nerve root irritation, foraminal stensois, or cord compression, but is also used to mean the effects of central sensitisation and central disinhibition, where the patient experiences widespread pain, not necessarily attributable to any pathology or correlated with its severity. The latter can be relevant in OA pain and should be picked up during examination and initial assessment (or of course may occur as the disease progresses). Gabapentin is therefore indicated when allodynia and hyperalgesia are present or where the pain and suffering is not being controlled by other means. There are various theories as to how this drug works; the most commonly quoted is that it blocks calcium channels thereby decreasing the excitability of nerve cells, slowing transmission, but there may also be some noradrenergic effects via descending inhibition.
Side effects reported include sedation, vomiting and diarrhoea, excessive drinking (short lived), itch (at higher doses), but specifically, ataxia with stumbling and tripping, assumed to be associated with feelings of dizziness reported by human patients. Gabapentin is partially metabolised by the liver and eliminated by the kidneys in the dog, so minimal dosing is wise where there is renal or hepatic dysfunction. The recommended dosages are 5-10mg/kg bid, starting low and increasing if necessary. It appears to be safe to use alongside tramadol and NSAIDs. - Paracetamol...an NSAID, with no gastric activity, paracetamol can be safely used when there is gastric ulceration, and alongside other NSAIDs and glucocorticoids. The disadvantage of this drug is that it is not a very potent analgesic when used on its own. In humans, it is reported to be more potent in combination with other analgesics than when used alone. Its licensed combination contains codeine, and, for that reason, should really be avoided with tramadol, in case the patient is particularly sensitive to the opiate activity of both. It is contraindicated in liver dysfunction. The recommended dose is 10mg/kg bid.
- Amantadine...an NMDA receptor antagonist that is reportedly useful for controlling canine OA pain. However, it appears to be most useful in this regard alongside an NSAID or opiate, although there are anecdotal reports of success in its treatment of neuropathic pain as a sole agent. It would be indicated in uncontrolled OA pain, where there is evidence of central sensitisation, and where gabapentin/tramadol is not tolerated or not working. Side effects are sedation and vomiting/diarrhoea. Recommended doses are 2-4mg/kg sid.
- Amitryptline... a tricyclic anti-depressant (TCA) which is an effective analgesic for both neuropathic and nocioceptive pain in humans, when used at lower doses than for emotional disorders. However, it has not been assessed in canine OA. It has been dubbed a multimodal analgesic on its own because of its widespread actions via descending inhibitory pain control, regulation of sleep, anxiolysis, and antihistamine effects. It has more noradrenergic activity than other TCAs and therefore is considered superior to them for pain relief in humans. The SSRIs (specific serotonin reuptake inhibitors such as fluoxetine) have not been found at all useful for analgesia.
Amitryptline requires competent hepatic and renal function for elimination. In humans it causes drying of the mucous membranes and therefore should perhaps be used with caution in KCS. It is dysrhythmogenic in overdose and should be used with extreme caution in patients with dysrhythmias. Because of these cautions, it tends to be used less frequently than tramadol and gabapentin, although time and experience may also inhibit the use of these in certain circumstances. Side effects of amitryptline include transient vomiting and diarrhoea, sedation, and occasionally, changes in behaviour (disinhibition).
ACUPUNCTURE
Acupuncture is being used increasingly in the treatment of OA pain in humans and animals (Figure 6). There are now systematic reviews to support the use of acupuncture in OA of the human knee, but there are few useful studies in the canine field. Currently, acupuncture should be thought of as primarily providing pain relief, rather than an anti-inflammatory action, although there is an emerging suggestion that the treatment of pain may modify disease. Until this is more firmly grounded, it is sensible to use acupuncture as an adjunct to treat OA, rather than a sole treatment if an active inflammatory process is suspected. Acupuncture triggers and enhances potent analgesic pathways in the body via the stimulation of (primarily) A delta (or Type II and III) fibres in skin and muscle. This stimulation causes inhibition of C fibre pain in the dorsal horn and release of beta-endorphins, noradrenaline and serotonin, amongst other neurotransmitters, from the brain.17
(Figure 6). There are now systematic reviews to support the use of acupuncture in OA of the human knee, but there are few useful studies in the canine field. Currently, acupuncture should be thought of as primarily providing pain relief, rather than an anti-inflammatory action, although there is an emerging suggestion that the treatment of pain may modify disease. Until this is more firmly grounded, it is sensible to use acupuncture as an adjunct to treat OA, rather than a sole treatment if an active inflammatory process is suspected. Acupuncture triggers and enhances potent analgesic pathways in the body via the stimulation of (primarily) A delta (or Type II and III) fibres in skin and muscle. This stimulation causes inhibition of C fibre pain in the dorsal horn and release of beta-endorphins, noradrenaline and serotonin, amongst other neurotransmitters, from the brain.17
PHYSIOTHERAPY
Physiotherapy is increasingly used in small animals for a wide range of problems. Like acupuncture it is an emerging field in veterinary medicine, with limited research to demonstrate efficacy in the veterinary species. However, it should be remembered that lack of evidence of efficacy is not evidence of lack of efficacy. Additionally, there are complex reasons regarding the assessment of any physical therapy (including acupuncture), that are outside the remit of this article, as well as simpler reasons regarding funding, that are limiting progress in these fields.
Meanwhile, combinations of therapies including cryotherapy (for acute inflammation) and the application of heat (when acute inflammation has resolved); range of motion exercises; hydrotherapies, including swimming and underwater treadmill walking (Figure 7); electrical nerve stimulation, and active exercise, appear to be the most helpful in rehabilitating, or palliating, patients with OA. Exercises include sit-to-stands (Figures 8a, b,c), “dancing”, shaking hands, step climbing and lead walking. Such exercises may be best designed and demonstrated by a qualified physiotherapist, although the simpler strategies can be easily learned and demonstrated. Beware of forcing or encouraging animals with severe pain to engage in such activities without the benefit of sufficient analgesia.

B = Bodyweight
The overall results of various studies on obesity demonstrate that weight loss is beneficial for dogs with OA. This is not just about a decreased load on the joints, although this is relevant in gross obesity. The presence of excess fat itself causes more inflammation and pain. Although weight loss appears to be a simple equation where giving fewer calories results in weight loss, this overlooks the important and complex relationship between the provision of food and the provision of affection. Weight reduction clinics should focus on the individual difficulties of each owner and pet in the struggle to lose weight, withhold judgment, encourage positive techniques (such as the novel food delivery systems described below) and avoid pejorative terms related to obesity. Some patients are underweight and poorly muscled, which, in some cases will be related to other disease processes that must be addressed. In other cases, specific nutritional measures may need to be discussed.
C = Control
Control of complications begins with owner awareness. Owners should be made aware, preferably in writing, of the side effects to which they should pay attention, and the measures they should take. The presence of these side effects should be checked at each consultation by questioning (not relying on the owner to volunteer the information) and examination.
Routine or specific blood work should be regularly carried out, especially in geriatric, or especially vulnerable, animals. Minimal dosing should be employed where possible, whilst ensuring that analgesia is sufficient.
Control of comfort and resources... improving environment and comfort will improve the patient’s quality of life, potentially impacting on their pain. Some of these measures are commonsense, some a little more unusual:
- The choice of bed should be driven by the patient’s preference, not by what is appealing to the owner. Does the dog like to lie flat out, or curled, or against something solid? Does it prefer a firm place to lie or a soft one – or would it be better with a choice?
- Some dogs become less comfortable travelling in cars when they have OA because of the movement and will try to brace themselves in a foot well or against a solid surface. Create a padded area such that they can do this easily. Ramps can be helpful, but be aware that some dogs may need training first, with the ramp flat and then increasingly tilted. Beware – dogs who have a wide gait, for example dogs with elbow OA often abduct the leg as they walk and may fall off a ramp of normal width. The ramp should have grips and be robust and not make a noise when the dog steps onto it.
- Dogs that pull on leads may continue to hurt themselves, so judicious use of headcollars or nonpulling harnesses can be helpful. Avoid harnesses that run under the axillae because they have a tendency to cause dogs to abduct their elbows.
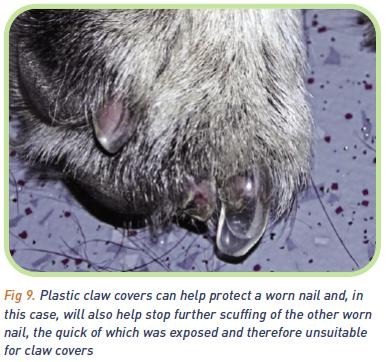 If dogs are scuffing their paws and nails, boots may be helpful, although often difficult to fit. A novel approach may be to fit plastic claw covers before the quick is exposed and allow the claw cover to take the wear (Figure 9; www.softpaws.net). The use of nonslip rugs on wooden or laminate floors will assist dogs who have difficulty moving about on such surfaces. Raising the feeding dish may also be helpful to stop dogs having to bend to reach their bowl. Be aware of the substrates on which the dog is walking; restricted exercise usually means “round the block” on concrete - try to encourage owners to keep the dogs to grass and/or firm sand, where possible.
If dogs are scuffing their paws and nails, boots may be helpful, although often difficult to fit. A novel approach may be to fit plastic claw covers before the quick is exposed and allow the claw cover to take the wear (Figure 9; www.softpaws.net). The use of nonslip rugs on wooden or laminate floors will assist dogs who have difficulty moving about on such surfaces. Raising the feeding dish may also be helpful to stop dogs having to bend to reach their bowl. Be aware of the substrates on which the dog is walking; restricted exercise usually means “round the block” on concrete - try to encourage owners to keep the dogs to grass and/or firm sand, where possible.- Mental stimulation can be important for dogs that have had their resources markedly reduced (no walks, no play, less food!) and may also have an impact on pain by literally taking their mind off it. If walks are restricted, make sure that they are varied regularly. Change play to hide and seek for toys or food rather than running after a toy. Use food balls, or a plastic bottle with holes punched in it, to allow the dog to take longer eating the same food ration, or hide dry food in a box with shredded paper. The food ball can also be used in hide and seek games or as a reward for new behaviours or tricks. Dogs that are very people orientated can even go visiting or have their favourite people to visit them!
- Touch is a potentially powerful tool, releasing neurotransmitters related to mood, analgesia and bonding, assuming the touch is of a positive kind.18 Simple tickling, or stroking the abdomen and chest, or a flat-handed massage, may be enjoyed (not endured) by the patient. Discourage the owner from using their fingers to dig into the muscles. In a flat-handed massage the palm or “heel” of the hand is placed in contact with the skin and the skin and hand moved together over the underlying tissues. Much gentler contact, or avoidance of this altogether, would be indicated if the patient demonstrates allodynia.
Many of these techniques give the owner a sense of control of their pet’s problem. Including simple physiotherapy exercises, when appropriate, can also help. If owners do not feel as though they have anything to contribute, they may search for alternative, and possibly inappropriate, treatments, or become disillusioned with what their practice has to offer.
D = Disease modifying agents
These are the “Holy Grail” of OA treatment and are included more in hope for the future than in expectation of anything definitely effective in the present. There are mixed results for studies into polysulphated glycosaminoglycans, pentosan polysuplhate and hyaluronan; however, anecdotal reports continue to be positive and some patients appear to benefit greatly. As previously mentioned, the jury remains out on gluco samine and chondroitin supplements and green-lipped mussel preparations, but there is more promising evidence on omega 3 based diets. Surgery can also be used to modify OA and reduce pain; techniques include arthrodesis, prostheses (hip, elbow and stifle), amputation and femoral head excision.
E = Exercise
Exercise is important to improve muscle strength, cardiovascular fitness, reduce weight, and limit stiffness and muscle wastage. It is also important for most dogs’ wellbeing and mental stimulation, as well as for most owners’ continuing enjoyment of their pet. Therefore, whilst complete rest is important in the acute phase of the disease, or during an acute flare up, a gradual increase in exercise is important once that phase has passed. It is also important to give owners more than a vague direction. “Five minutes lead walking on flat ground three times daily” leaves little room for misinterpretation and owners can then see how their pet is progressing as the allowance increases. Use realistic measures rather than random restrictions, for example: the owner may need to determine the length of walk the dog can have after which it is not stiff or lame when rested, or the length of walk which means it is not slow on the return journey, or does not become lame during the walk.
SUMMARY
This all takes time and there are no short-cuts. However, this does not have to all be achieved in one ten or fifteen minute consultation. Additional information and additional advice can be added at each consultation so that the owner takes something new away with them every time; a new exercise, a reduction in medication, a new game to play, a new physiotherapy technique, some additional information about chronic pain.
After all, OA is a life-long condition that frequently changes its presentation - who would expect to have that all wrapped up after just one consultation?
This article was kindly provided by Merial, makers of Previcox:


References
1. Simons DG, Travell JG, Simons PT. In: Travell and Simons’ Myofsacial Pain and Dysfunction: The Trigger Point Manual. Volume 1, Upper half of the body. Williams and Wilkins, Baltimore,1999.
2. Gerwin RD. Neurobiology of the myofascial trigger point. Ballieres Clinical Rheumatology 1994; 8(4): 747-762.
3. Kellgren JH. Observations on referred pain arising from muscle. Clinical Science 1938; 3: 175-190.
4. Lindley S, Cummings M. Essentials of western veterinary acupuncture. Blackwells, Oxford. 2006.
5. Lamont LA, Tranquilli WJ, Grimm KA. Physiology of pain. Veterinary clinics of North America small animal practice 2000; 30(4): 703-728.
6. Mathews KA. Update on management of pain: Neuropathic pain in dogs and cats: If only they could tell us if it hurts. Veterinary Clinics of North America Small Animal Practice 2008; 38(6): 1365-1414.
7. Sluka KA, Jordon HH, Westland KN. Reduction in joint swelling and hyperalgesia following post-treatment with a non-NMDA glutamate receptor antagonist. Pain 1994; 59: 95-100.
8. McCarthy G, O’Donovan J, Jones B and others. Randomised double-blind, positive controlled trial to assess the efficacy of glucosamine/chondroitin sulfate for the treatment of dogs with osteoarthritis. The Veterinary Journal 2007; 174: 54-61.
9. Moreau M, Dupuis J, Bonneau NH and Desnoyers M. Clinical evaluation of a nutraceutical, carprofen and meloxicam for the treatment of dogs with osteoarthritis. The Veterinary Record 2003;152: 323-329.
10. Holton L, Reid J, Scott E M and others. Development of a behaviour based scale to measure acute pain in dogs. The Veterinary Record 2001; 148: 525-531.
11. Wiseman-Orr M L, Nolan AM, Reid J and Scott EM. Development of a questionnaire to measure the effects of chronic pain on health related quality of life in dogs. The American Journal of Veterinary Research 2004; 65: 1077-1084.
12. Yeates J and Main D. Assessment of companion animal quality of life in veterinary practice and research. The Journal of Small Animal Practice 2009; 50: 274-281.
13. Webster J. In: Animal Welfare; Limping towards Eden. Blackwell Publishing, Oxford 45, 2005.
14. Carmichael S. Putting theory into practice - best practice management for osteoarthritis. 9th Annual Symposium, Geneva, 2005.
15. Lascelles BD, King S, Roe S and others. Expression and activity of COX-1 and 2 and 5-LOX in joint tissues from dogs with naturally occurring coxofemoral joint osteoarthritis. The Journal of Orthopaedic Research 2009; 27(9): 1204-1208.
16. Veiga AP, Duarte IDG, Marcelo NA and others. Prevention by celecoxib of secondary hyperalgesia induced by formalin in rats. Life Sciences 2004; 75: 2807-2817.
17. Bowsher D. Mechanisms of acupuncture. In: Filshie J, White A, editors. Medical Acupuncture: A Western scientific approach. Churchill Livingstone, Edinburgh, 69-82, 1998.
18. Odendaal J. Pets and our Mental Health: The why, the what, and the how. Vantage Press: New York, 2002.

