Show all small animal articles
Spinal Injury
Mark Bush MA VetMB CertSAS MRCVS - 06/12/2014
Spinal Injury
INTRODUCTION
Spinal pathology is seen as a result of a vast number of disease processes. Whilst in many cases detailed investigation and definitive surgical or medical treatment may be beyond the remit of the primary clinician, the initial assessment, adequate stabilisation of the patient and a thorough neurological examination will inform the clinician and owner and can influence the outcome of the case. This article aims to provide a straightforward, practical overview to the spinal patient. There is a brief summary of spinal anatomy and function and a focus on two commonly encountered causes in dogs, cord contact injuries due to intervertebral disc disease and trauma, but the principles described apply to any spinal pathology. It will then look at the assessment, investigation, treatment options and prognoses of spinal cases. For the purposes of the article, the term trauma will apply to injuries occurring to the spine following application of external forces, such as a road traffic accident or a fall from a height. Spinal cord contact injuries include injuries to the spine as a result of a trauma or intervertebral disc disease.
INTERVERTEBRAL DISC DISEASE (IVDD)
Each vertebra is separated by an intervertebral disc. Intervertebral discs comprise an eccentrically positioned nucleus pulposus surrounded by concentric fibrocartilaginous rings of the annulus fibrosus (Figure 1). Initially the nucleus pulposus is a semi-liquid gel, helping the disc to act as a shock absorber between the vertebrae. Over time this material degenerates as the disc dehydrates. In chondrodystrophic breeds, the nucleus can become mineralised. In other breeds the nucleus undergoes fibroid degeneration. The eccentric position of the nucleus pulposus means the dorsal aspect of the annulus is thinner than the lateral or ventral aspects; this is of significance in the pathology of disc disease1.
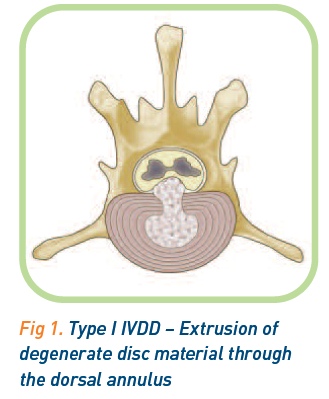 Type I IVDD. The extrusion of degenerate disc material through the dorsal annulus.
Type I IVDD. The extrusion of degenerate disc material through the dorsal annulus.
This is commonly seen as an acute presentation in chondrodystrophic breeds following the movement of the mineralised nucleus through a tear in the dorsal annulus into the spinal canal1 (Figure 1).

Type II IVDD. The protrusion of the intact dorsal annulus following collapse of the intervertebral disc space.
This is usually seen as a more gradual onset of disease in nonchondrodystrophic breeds. The degenerate nucleus no longer supports the disc space, the resultant collapse of the disc space causes the annulus to bulge dorsally, compressing the cord1 (Figure 2).
Type III IVDD. An extrusion of healthy disc material 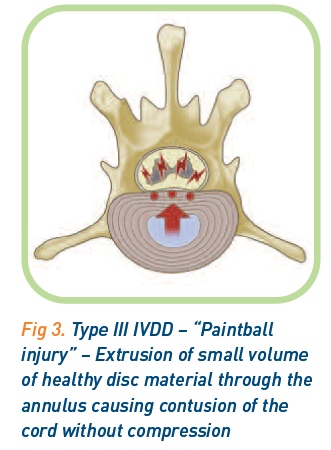 through the dorsal annulus.
through the dorsal annulus.
This is often seen in young animals following vigorous activity and is analogous to a ‘paintball injury’. A small portion of the healthy, hydrated, non-degenerate nucleus is fired through the annulus, hitting the cord at some speed, causing considerable concussive effects to the cord. The gel-like disc material immediately disperses, leaving no resultant compression2, 3 (Figure 3).
SPINAL CORD INJURY MECHANISMS
Contact with the cord causes injury via two mechanisms; contusion and compression of the cord.
Contusion or impact on the cord results in impairment of the perfusion of the affected area of the cord as the vessels supplying the spine are damaged. The system of autoregulation of spinal blood pressure is also impaired4,5 further exacerbating damage to the spine. Subsequently, a complex cascade of biochemical changes is triggered, resulting in the influx of sodium and calcium ions to the neurones, and release of free radicals. These ‘secondary’ changes occur rapidly, beginning within moments of the injury but peaking two to four days after the initial insult6. They cause neuronal cell death, apoptosis and an inflammatory reaction7.
The pathophysiology of compressive injury is less clearly understood. Its effects are somewhat dependant on the rate at which compression occurs8,9. Generally, compression leads to obstruction of the venous drainage and subsequent vasogenic oedema10. More chronic compression leads to ischemia, demyelination and death of the neurones.
Contact injuries can result in a spectrum of both mechanisms. For example, an acute extrusion of calcified disc material (Type I IVDD) will lead to both immediate contusion and compression. A slowly developing dorsal annulus protrusion (Type II IVDD) will cause more compression with some ongoing contusion, and extrusion of a low volume of healthy disc material (Type III IVDD) will cause contusion only.
INITIAL ASSESSMENT
Many spinal injuries will present as an acute paraplegic or paraparetic animal. Some will have had an obvious inciting cause, such as trauma, others will not be traumatic or may have a more insidious onset, over days or weeks. For animals seen following trauma, the priority of assessment is always the ABC of trauma: Airway, Breathing and Circulation, details of which are beyond the scope of this article.
Ensuring adequate oxygenation and establishing intravenous access with appropriate fluid rate will help to stabilise the cardiovascular system, maintain tissue oxygen perfusion, thus limiting the effects of shock. As mentioned above, cord injury causes a loss of autoregulation of the spinal cord blood supply, so shock will exacerbate damage to the cord. Similarly, where possible, a rush to anaesthetise the patient to begin a radiographic evaluation should be avoided until the animal’s cardiovascular system is adequately stabilised and supported because of the potentially deleterious effects of an anaesthetic on spinal blood pressure11.
Immobilising the patient on a spinal board will help to limit ongoing spinal damage due to unstable or compressive injuries. Ensuring adequate analgesia is important in all trauma patients.
Once the trauma patient is physiologically stable, it is important to assess the animal for orthopaedic injuries. Fractures or luxations of the appendicular skeleton may affect the animal’s ability to respond to the tests of the neurological exam and may mimic neurological injuries. Gentle palpation of the spine can be a sensitive indicator of potential spinal injury.
NEUROLOGICAL ASSESSMENT
The fundamentals of the neurological exam are firstly to establish that the problem is indeed neurological. Next it will give an idea as to where along the length of the cord the lesion is located (neurolocalisation) and how severe the grade of injury is. The exam will guide further investigations, help to formulate a differential list and provide an initial prognosis prior to further investigation.
Movement and mentation
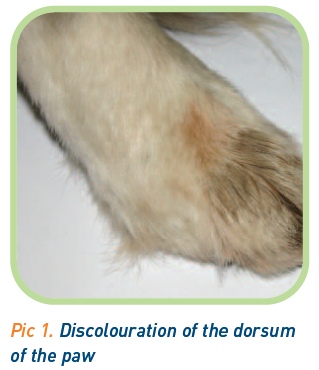 The first stage of a neurological exam is to watch the animal move. Clearly this is not advised if spinal instability is suspected or if movement is precluded by discomfort. However, if the case is ambulatory either with or without support, look for evidence of voluntary movement and coordination of the limbs. Does there appear to be adequate muscle strength? Are just the pelvic limbs affected, is the condition lateralised, or even just affecting a single limb? Can you hear the nails being scuffed? An animal with chronic proprioceptive deficits may have a discoloured dorsal aspect to its paws (Picture 1). If an animal has a cervical spinal lesion, its movement will often be altered as the animal’s head and neck is held rigid.
The first stage of a neurological exam is to watch the animal move. Clearly this is not advised if spinal instability is suspected or if movement is precluded by discomfort. However, if the case is ambulatory either with or without support, look for evidence of voluntary movement and coordination of the limbs. Does there appear to be adequate muscle strength? Are just the pelvic limbs affected, is the condition lateralised, or even just affecting a single limb? Can you hear the nails being scuffed? An animal with chronic proprioceptive deficits may have a discoloured dorsal aspect to its paws (Picture 1). If an animal has a cervical spinal lesion, its movement will often be altered as the animal’s head and neck is held rigid.
Proprioception
Proprioception is the body’s conscious awareness of its own movement and position of its limbs in space. Sensory receptors in the limbs detect movement and the position of the limbs and relay that information to the brain. Proprioception is of use in the assessment of our patients because this information must travel from the limb to the brain via spinal pathways (the dorsal and lateral funiculi). Therefore, presence or absence of proprioception will allow us to assess the integrity of the entire spine from the input of the limb being tested, to the brain.
Proprioceptive information must be transmitted rapidly to allow for adjustments in movement to take place quickly.  The speed of nerve conduction is determined firstly by diameter of the nerve fibre; the larger the diameter, the faster the conduction. Secondly, by the presence of a myelin sheath around the fibre; myelinated fibres conduct impulses significantly quicker than non-myelinated fibres. Proprioceptive fibres are large and myelinated. This is important in spinal injury as larger diameter fibres are also less resistant to damage than small diameter fibres; myelinated fibres are also more susceptible to damage than non-myelinated fibres.
The speed of nerve conduction is determined firstly by diameter of the nerve fibre; the larger the diameter, the faster the conduction. Secondly, by the presence of a myelin sheath around the fibre; myelinated fibres conduct impulses significantly quicker than non-myelinated fibres. Proprioceptive fibres are large and myelinated. This is important in spinal injury as larger diameter fibres are also less resistant to damage than small diameter fibres; myelinated fibres are also more susceptible to damage than non-myelinated fibres.
In addition, the location of the spinal pathways at the periphery of the spinal cord means these fibres are in a more vulnerable position in the event of injury (Figure 4). For these reasons proprioceptive function is a very sensitive indicator of spinal cord injury; it will be the first neurological deficit that becomes apparent and is usually the last to resolve during recovery.
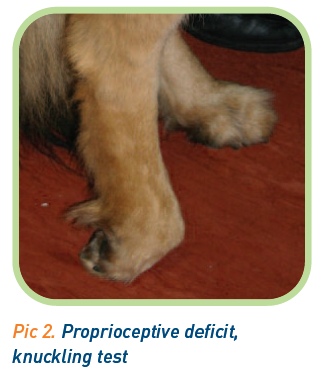
Proprioception can be tested in a number of ways. Commonly the ‘knuckling’ test is used. This will test the proprioceptive function of the distal limb. The animal’s paw is turned over such that the dorsum of the paw contacts the ground. The animal should reposition the paw immediately. Deficits can be very obvious, with no attempts made to correct the paw placement, or more subtle, with the recovery taking some time. This test will often need to be repeated several times. It is vital that the animal’s weight is fully supported when performing this test as weakness or injury unrelated to spinal cord disease can give false findings (Picture 2).
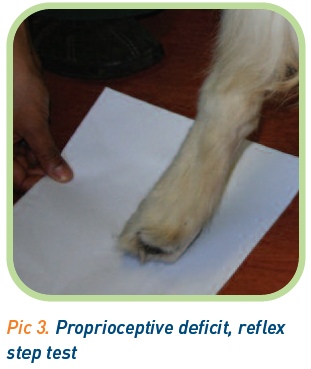
The ‘paper slide’ or reflex step test will examine the proprioception of the more proximal aspect of the limb. The paw is placed on a piece of paper, which is then drawn away from the animal. The normal animal should reposition its limb without delay (Picture 3).
Further tests may be required where subtle proprioceptive deficits are suspected, or when the patient is a cat. Wheelbarrow walking the patient on their thoracic or pelvic limbs, hemiwalking on the limbs of one side or hopping the animal on the limb under examination should demonstrate coordinated, regular movement of the normal limb(s) in attempts to support the animal’s body weight. Finally, tactile placing response is tested by covering the eyes of the animal whilst lifting it towards a table-top. As the dorsal aspect of the paw touches the underside of the table, the animal should respond by moving its limb to place it on the table. These tests are normally reserved for those cases where proprioception is equivocal.
Don’t attempt to wheelbarrow or hop an animal where spinal injury is suspected due to trauma or spinal instability.
The proprioceptive function of each limb should be assessed in turn. This will help to localise deficits to pelvic limbs only, all four limbs, unilateral lesion or a monoparesis.
Normal proprioception does not preclude a low-grade spinal cord injury, sometimes pain will be the only evident clinical sign. However, where an animal is exhibiting clinical signs consistent with a severe cord injury – for example, it is non-ambulatory but proprioception is present – the diagnosis should be reconsidered.
A number of other conditions can mimic spinal injury.
A small selection of non-spinal causes of ‘spinal’ presentations:
• Bilateral cruciate ligament rupture
• Myesthenia gravis
• Hypoadrenocorticism
• Hypoglycaemia
• Peripheral neuropathies
• Prostatic disease
• Botulism
Having established there is a pathway problem at a point somewhere between the proprioceptively deficient limb and the brain, its location can be further refined by assessing spinal reflexes, muscle tone, strength and mass.
Spinal reflexes
The canine spine comprises 31 segments of interest; 8 cervical segments, 13 thoracic segments, 7 lumbar and 3 sacral, each of which supplies a lower sensory and motor spinal nerve. The spine can be functionally divided into 5 areas (Figure 5): a corresponds to segments C1-C5, b C6-T2, c T3-L3, d L4-S1 and e S1-S3.
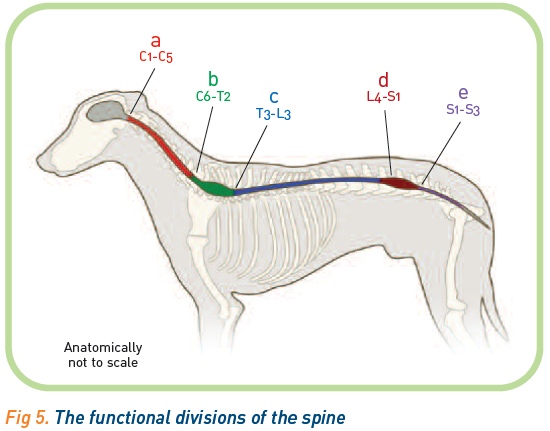
Areas b and d represent the concentration of cell bodies of lower motor neurones in the grey matter supplying the thoracic limbs (cervical intumescence C6-T2, b) and the pelvic limbs (lumbar intumescence L4-S1, d). Regions a (C1-C5) and c (T3-L3) mainly comprise the upper motor neurone pathways connecting the cell bodies in the brain and the lower motor neurone; the white matter. Area e corresponds to the sacral segments of the spine (S1-S3). This area innervates part of the sciatic nerve, the bladder and anal sphincter and tail.
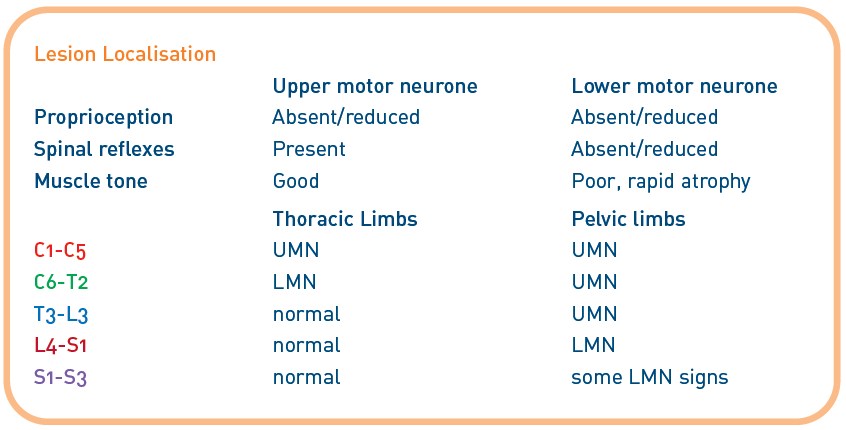
Lower motor neurones are responsible for effecting motor activity; impairment of their function will lead to loss of reflex activity, loss of muscle tone, and rapid muscle atrophy. Upper motor neurones have an inhibitory function on the lower motor neurone. Injury to the upper motor neurone removes the inhibitory effect on the lower motor neurone, and increases reflex activity and muscle tone.
Whilst performing the spinal reflexes, a note should be made of muscle tone, strength and mass. A detectable resistance to passive movement of the limb indicates the presence of muscle tone.
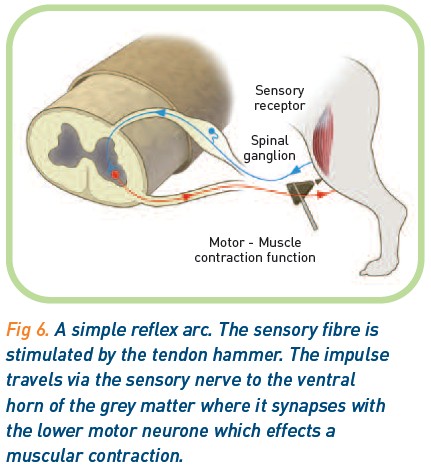
Spinal reflexes examine simple reflex arcs (Figure 6) . Sensory receptors in the muscle or tendon detect the stimulus and synapse with motor neurones in the spinal cord. The motor neurones cause the appropriate muscles to contract. No spinal pathways to the brain are involved. Reflexes can be most simply classed as present or absent. They are generally easier to perform with the animal laid down on its side.
Pelvic limb spinal reflexes that can be most reliably tested are the patellar reflex and flexor withdrawal. Patellar reflex is elicited by striking the straight patellar ligament briskly with a tendon hammer or suitable alternative. A reflex extension of the stifle is a positive result. Occasionally ongoing tremor of the muscle (a clonic response) is seen, which can indicate chronicity of the lesion. Absence of response suggests a problem affecting spinal segments supplying the femoral nerve (L4-L6), or less likely, the nerve itself.
Flexor withdrawal is elicited by squeezing the toes of the paw. A strong contraction of the stifle and, in particular, the hock should be seen regardless of whether or not the animal is making a conscious response. Absence of a withdrawal response indicates problems with segments of the cord corresponding to the sciatic nerve (L6/7-S1), or the nerve itself.
DON’T confuse unconscious flexor withdrawal with conscious deep pain response (see below for a description of deep pain assessment).
Thoracic limb spinal reflexes are much less dependable. They are not always present, reliable or reproducible in normal animals. Extensor carpi radialis, biceps brachii muscle reflexes may be tested. Flexor withdrawal of the thoracic limbs can be a help in assessing for cervical intumescence lesions; however, even this test is not wholly accurate in distinguishing lesion in site a from b12. An assessment of muscle tone of the limbs may be of some use. Definitive distinction between lesions in site a from b is not always possible by clinical exam alone12.
Neck pain is very characteristic of cervical spinal disease but for some very mildly affected tetraparetic animals, close examination of the gait and coordination of the movement during hopping or wheelbarrow walking of the thoracic limbs may be the only clue that the cervical spine is affected.
Where spinal reflexes and muscle tone and strength are absent or poor, this will correspond to a lesion in the intumesence of that limb (b, d or e). Where spinal reflexes are present and muscle tone and strength is good, the lesion will usually correspond to a lesion of the pathways (a or c).
Lesions affecting the sacral segments (S1-S3) will typically have less dramatic effects on limb use; however, as a component of the sciatic nerve is supplied by S1, some deficits may be seen. The animal will normally be ambulatory, although discomfort may preclude normal activity. Pelvic limb proprioception and flexor withdrawal reflex may be diminished. Sensation to the tail can be reduced. Notably the anal tone is often poor, and the sphincter will not contract in response to perianal stimulus. More severely affected animals can become faecally and urinary incontinent.
Grading the severity of the lesion
The severity of spinal injury can be inferred from the deficits detected and can be graded accordingly. As mentioned above, the diameter of the nerve fibres within the cord will affect their susceptibility to injury. Motor fibres are of intermediate diameter. Pain sensation is conducted by small diameter myelinated and unmyelinated fibres and hence is the least susceptible to injury. Furthermore, the pathways that relay pain perception to the brain are centrally located within the cord and cross from one side of the cord to the other at multiple points along the spine, so damage to the spine must be very profound to abolish pain sensation. Bladder function (S1-S3) is relatively resistant to spinal injury and most cases with bladder dysfunction will be paraplegic. An accurate assessment of bladder function can be difficult in acute presentations. Injury to the spine cranial to S1-S3 will result in an ‘upper motor neurone bladder’ where urine is retained and expression of the bladder is difficult; injury to segments S1-S3 and the peripheral nerves will result in a ‘lower motor neurone bladder’ and an incontinent animal.
Grade 1 - Pain only. Ambulatory with no other deficits noted.
Grade 2 - Tetra or Paraparesis. Ambulatory with loss of proprioceptive function, weakness and ataxia.
Grade 3 - Tetra or Paraparesis. Non-ambulatory, but evidence of voluntary movement in the limbs.
Grade 4 - Tetra or Paraplegia. Non-ambulatory, with no evidence of voluntary movement in the limbs, deep pain sensation present. +/- Urinary function.
Grade 5 - Paraplegia. As above, with no evidence of deep pain sensation.
Paraparesis is weakness and ataxia of the limbs. Paraplegia is defined as a loss of motor function.
This grading system is of use in describing the degree of injury; however, as a prognostic indicator, the most significant factor is the presence or absence of deep pain sensation.
Deep pain sensation
Deep Pain Sensation need not be assessed in animals that can walk. Except in very unusual presentations, it will always be present.
Deep Pain Sensation (DPS) can be commonly misinterpreted. The test requires increasingly forceful pressure to be applied to the bone of the distal pelvic limb, typically a phalanx. DPS is relayed to the spine via the sensory neurones, as for the flexor withdrawal reflex arc. In cases where the arc is still functioning, unconscious withdrawal will still occur regardless of the severity of the lesion. For deep pain sensation to be classed as present, the information must be transmitted to the brain (via the spinaothalamic and propriospinal pathways) and the animal must make a repeatable, conscious response to the stimulus (vocalising, turning towards the stimulus, attempting to bite the examiner etc). A conscious response is the significant factor. An animal with a lesion in the lumbar intumescence may not withdraw its foot in response to a painfulstimulus, but if it consciously reacts to the stimulus, DPS is present. Conversely, an animal that withdraws its limb but is not consciously aware of the stimulus has no deep pain sensation. The presence or absence of deep pain sensation is the most significant indicator of prognosis13-16.
OTHER ASSESSMENTS
Panniculus response, or Cutaneous Trunci Reflex
The panniculus response is of limited practical value in assessment of spinal injuries. Stimulation of the skin of one side of the trunk sends an impulse to the spine via sensory fibres of that area of skin (dermatome). The impulse passes cranially in the cord to synapse with motor neurones present segments C8-T1 of the cord. The motor neurones affect a bilateral twitch of the cutaneous trunci muscle. The response may be lost if the cord is damaged at a point cranial to the input of the sensory fibre. It must be borne in mind that the dermatomes project caudally from the spine such that the L1 dermatome extends to the tuber coxae, so lesions caudal to the L1 segment will result in a normal reflex bilaterally. The reflex may be completely absent in some lesions affecting the C8-T1 segments of the spine, and will be unilaterally absent following brachial plexus avulsion.
The reflex may occasionally assist in neurolocalisation if a myelogram is equivocal or MRI is unavailable.
Schiff-Scherrington Sign – loss of ascending tract inhibition
Cells of the dorsolateral grey matter in spinal segments L1-L7 project cranially and provide inhibitory inputs to the neurones supplying the thoracic limbs. Damage of the cord at segments L1-L7 can lead to a loss of the inhibitory input, and thus increased muscle tone. This presents as increased thoracic limb extensor tone; the limbs are held rigidly outstretched. At first glance this may be confused with an injury to cord segments C1-C5, as described above; however, the two can be readily distinguished by the presence of normal proprioceptive function in the thoracic limbs in cases exhibiting Schiff-Scherrington signs. It was once thought that loss ascending tract inhibition was associated with a poor prognosis, but this is no longer the case17.
Differential diagnoses
The history of the presentation, signalment of the animal and neurolocalisation of the lesion will help to plan a differential diagnosis list.
The DAMNIT-V classification will help to identify potential aetiologies. More exhaustive differential diagnoses lists should be consulted. The next step of investigation is usually imaging the spine.

IMAGING
Plain radiography
Plain radiography is inexpensive and virtually ubiquitous in small animal practice. It is valuable in the identification of spinal fractures or luxations, discospondolyitis, congenital abnormalities and some neoplasia. It can provide evidence to support a diagnosis of disc disease and degenerative changes. Its limitations are its lack of sensitivity in imaging soft tissues, and the fact it is restricted to two dimensions of imaging. Radiography is usually performed under deep sedation or general anaesthesia when the natural muscular splinting of the spine is lost, so great care must be taken to ensure the patient’s spine is kept in neutral alignment throughout the procedure, and any movements are controlled and kept to a minimum. As above, the cardiovascular system of any spinal injury patient must be stabilised and supported prior to general anaesthesia for radiography.
Plain radiography may provide a diagnosis in some cases; however, most will require further imaging to establish the diagnosis. Typically this will be one of myelography, computer tomography (CT scan) or magnetic resonance imaging (MRI).
Myelography
Myelography involves the intradural injection of a radiopaque, non-ionic contrast medium at either the lumbar or cisternal space, then subsequent radiography. The passage of contrast along the subarachnoid space will be attenuated at the area of a compressive lesion. Myelography can identify the site, and supply information as to the likely aetiology of the disease; however, its findings are not always definitive as the attenuation of contrast may occur over a large area of the spine, making lesion localisation difficult. Myelography is inexpensive and readily available. It can allow quite rapid assessment of the entire cord. Disadvantages of myelography include a risk of inducing seizures or exacerbating neurological damage18,19. It is unable to image the parenchyma of the cord and is very limited in the resolution of other soft tissues. Like radiography, Myelography is restricted to two dimensional images.
Computed Tomography
This modality uses multiple radiographic projections to provide more detailed images of the spine. Its significant advantages over radiography include very rapid imaging of the entire spine and that it allows imaging in the third dimension, giving transverse or axial sections of the spine. Its disadvantages are principally poor soft tissue resolution and lack of availability.
Magnetic Resonance Imaging
MRI is accepted as the current gold standard for imaging the spine20. MRI provides detailed images of both soft tissue and bone allowing an accurate assessment of vertebrae, paraspinal soft tissue, intervertebral discs and the spinal cord parenchyma in multiple planes and along the entire length of the cord. Its limitations are primarily those of cost and availability. The length of time taken to perform a scan is greater than that of CT. Continuing advances in this field of technology mean the impact of these factors is diminishing. (Pictures 4 & 5)
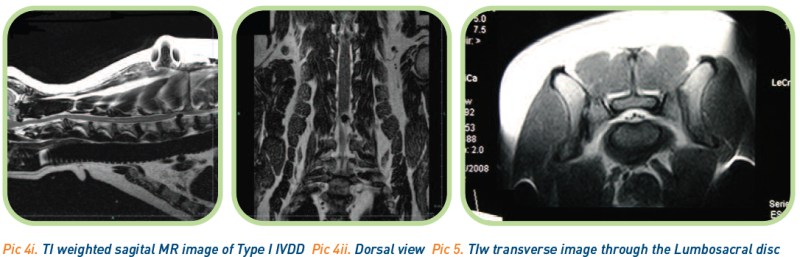
All imaging modalities have a learning curve for accurate interpretation. The wealth of detail produced by MRI scans means incidental and artefactual findings are more commonplace than those seen with radiography20.
CSF taps
CSF taps are generally indicated where a compressive lesion is not evident. They are of most use in differentiating infectious from non-infectious causes of inflammatory cord disease. They require general anaesthesia and can be performed at the lumbar or cisterna magna sites. Details of the procedure are widely published17.
TREATMENT AND PROGNOSIS
Treatment of spinal cord contact injury can be divided into conservative or surgical options.
As a general rule surgery is likely to offer a more rapid and reliable return to normal limb function. Low-grade injuries, where pain is the only clinical sign may be managed conservatively; however, where more severe neurological deficits are present, surgery is recommended17. Where an animal has lost deep pain sensation as a result of IVDD, rapid surgical decompression is essential16.
Intervertebral disc disease
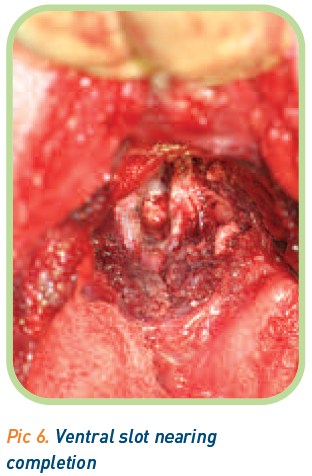
Surgical treatment for extruded degenerate disc material (Type I IVDD) typically involves the burring away of the vertebral lamina to access the cord and remove the compressive disc material. This will usually be via a hemilaminectomy for injuries of the thoracolumbar spine, a ventral slot for cervical spinal injuries (Picture 6) or a dorsal laminectomy for a lumbosacral location. In cases where deep pain sensation is present, the prognosis for a return to normal limb function following a thoracolumbar disc extrusion is approximately 80-97%13,14,16,21,22. Once deep pain sensation has been lost, the prognosis drops to 60%16. It was previously thought that injuries to the segments of spine making up the intumesences (C6-T2 and L4-S1) carried less favourable prognoses that other areas of the spine23 because neuronal cell bodies would be damaged irreversibly. Recent evidence suggests this is not the case24,15.
Surgical management of protruding dorsal annulus (Type II IVDD) in the thoracolumbar spine is less satisfactory. The annulus may be approached via a hemilaminectomy and resected, before stabilising the spine with pins and cement or a plate22. More recently the lateral corpectomy has been described in the management of type II IVDD25. The prognosis for type II IVDD is less favourable than type I22, possibly as a consequence of the fact the longer term compressive injury is less reversible than short-term concussive effects26.
Management of cervical spine type II IVDD is controversial; surgical management involves either, i) direct decompression of the cord via ventral slot27 or dorsal laminectomy28, or ii) indirect decompression by distraction and stabilisation of the affected disc space, such as with an intervertebral polymethylmethacrylate (PMMA) plug29, PMMA and screws30 – or recently the use of locking plates and a cortical allograft31. Choice of the procedure is dependant on factors such as the ‘traction response’ of the lesion, the number of sites affected and surgeon preference32,33. A review of the literature did not support the adoption of any one technique over the others; all techniques share a similar prognosis with approximately 75% making a return to useful limb function33.
As with all surgeries, complications can occur, including: potentially life-threatening intraoperative haemorrhage, post-operative surgical site or urinary tract infection, residual neurological deficits including faecal incontinence and recurrence of disc disease at the original site or a second disc.
Conservative management of IVDD may be chosen where costs preclude surgery or the animal is not exhibiting neurological deficits. Restricting the animal’s activity is the mainstay of conservative treatment. The timescale for this had not been rigorously defined, but a consensus of option suggests 2 weeks of strict exercise restriction such as cage rest, is appropriate as an initial trial. After this time, if the animal has shown improvement, the restriction should continue for a further 4 weeks. If the animal has not shown significant neurological improvement, if it has deteriorated or its pain is poorly controlled, surgical management should be recommended. A recent study showed that the prognosis for cases of cervical and thoracolumbar IVDD injuries managed conservatively is that approximately 49-55% return to normal limb function34,35. Owners should always be advised of the potential for sudden neurological deterioration in cases of IVDD managed conservatively and that up to one third of cases will suffer recurrence34-36. Conservative management of thoracolumbar type II IVDD results in approximately 50% of cases being euthanased by one year due to progression of the disease22. Use of a non-steroidal anti-inflammatory has been associated with an improved outcome in patients managed conservatively for suspected IVDD34,35.
For traumatic spinal injuries, early surgical decompression and stabilisation is the most appropriate management option. The prognosis is variable, depending on the type and severity of the injury; however, where a case has lost deep pain sensation secondary to a traumatic spinal injury, the prognosis is very poor16.
(Mis)use of corticosteroids in cases of spinal cord contact injury
The use of corticosteroids following spinal cord contact injury is a rather contentious issue. Thirty years ago, corticosteroid use, in particular dexamethsone, was accepted as the recommended first line treatment for acute spinal cord injury. The proposed mechanism of action was in a direct anti-inflammatory role. There is no evidence to support the efficacy of dexamethasone as a treatment for cord contact injury in dogs. Its use may worsen neurological outcome37-41.
Methylprednisolone sodium succinate (MPSS) has been proposed and used following evidence of a neuroprotective function in vitro42,43 and recently in experimental animals44. These effects are independent of its corticosteroid effect, acting rather as a free radical scavenger and supporting spinal blood supply. However, studies in dogs have not demonstrated a clinical benefit following MPSS use24,45. Importantly, however, MPSS is associated with significant adverse effects, predominantly notable gastrointestinal pathology in up to 90% of dogs46-48. MPSS use may also have an adverse effect on recovery processes in the spine49. A review of the evidence of corticosteroid use in human emergency medicine for spinal cord injury found that complications may outweigh the benefits of the treatment effect50. As such the American Association of Neurological Surgeons and the Congress of Neurological Surgeons do not recommend MPSS as a standard treatment for acute spinal cord injuries38.
A number of potential therapies are under investigation for the treatment of spinal cord injuries; these include minicycline51, erythropoietin52, polyethylene glycol53 and stem cell therapy54. As yet none of the above treatments have proven clinical benefits.
Aftercare
It is important to make clients aware that anticipated recovery from spinal injuries can be a long, expensive and intensive process. Surgical intervention may remove the inciting cause of the injury, but the disease mechanisms will have resulted in ongoing cord damage. The spine can show remarkable restorative properties, both via direct recovery of the neuronal pathways affected and significantly by plastic ‘rewiring’ of the cord to reroute signals7. This essentially means nonambulatory animals need to learn to walk again. The time taken for recovery from IVDD is dependant to a large extent on the initial grade of injury, but owners could expect non-ambulatory dogs to begin to ambulate after approximately two weeks21,55. When deep pain sensation has been lost, and has not returned in 4 weeks, the prognosis for a return to normal limb use is very guarded and animals are likely to remain incontinent16. Rehabilitation includes massage and physical therapy, much of which can be performed by the owners. A structured professional therapy regime with a registered physiotherapist may be beneficial56,57, however owners should realise that they will need to take on much of the responsibility for the aftercare of the patient.
This article was provided by Merial, makers of Previcox®:


References:
1. Toombs JP, Waters DJ. Intervertebral disc disease. In: Textbook of Small Animal Surgery. 3rd edn. Slatter DH (ed). Philadelphia: Saunders 2002; 1193–6. 2. Yarrow TG, Jeffery ND. Dura mater laceration associated with acute paraplegia in three dogs. Vet Rec 2000; 146: 138–139. 3. Hay CW, Muir P. Tearing of the dura mater in three dogs. Vet Rec 2000; 146: 279–282. 4. Tator CH, Fehlings MG. Review of the secondary injury theory of acute spinal cord trauma with emphasis on vascular mechanisms. J Neurosurg 1991; 75: 15–26. 5. Ohashi T, Morimoto T, Kawata K, et al. Correlation between spinal cord blood flow and arterial diameter following acute spinal cord injury in rats. Acta Neurochir (Wien) 1996; 138: 322–329. 6. Olby N. Current concepts in the management of acute spinal cord injury. J Vet Intern Med 1999; 13: 399. 7. Jeffrey ND, Blakemore WF Spinal cord injury in Small Animals 1. Mechanisms of Spontaneous Recovery. Vet Rec 1999; 144: 407-413. 8. Tarlov IM, Klinger H, Vitale S. Spinal cord compression studies. II. Time limits for recovery after acute compression in dogs. Arch Neuro and Psych 1954; 70: 271-282. 9. Tarlov IM, Klinger H, Vitale S. Spinal cord compression studies III. Time limits for recovery after gradual compression in dogs. Arch Neurol and Psych 1954; 71: 588-598. 10. Bohlman HH, Emery SE. The pathophysiology of cervical spondylosis and myelopathy. Spine 1988; 13: 843-846. 11. Guha A, Tator CH, Rochon J. Spinal cord blood flow and systemic blood pressure after experimental spinal cord injury in rats. Stroke 1989; 20: 372–377. 12. Forterre F, Konar M, Tomek A and others. Accuracy of the withdrawal reflex for localization of the site of cervical disk herniation in dogs: 35 cases (2004-2007). J Am Vet Med Assoc. 2008; 232 (4):559-63. 13. Anderson SM, Lippincott CL, Gill PJ. Hemilaminectomy in dogs without deep pain perception. A retrospective study of 32 cases. Cal Vet 1991; 45: 24-28. 14. Black AP, Lateral spinal decompression in ht edog; a review of 39 cases. J Sm Anim Pract 1988; 29: 581-588. 15. Dhupa S, Glickman NW, Waters DJ. Functional 0utcome in dogs after surgical treatment of caudal lumbar intervertebral disk herniation. J Am Anim Hosp Assoc 1999; 35: 323–31. 16. Olby N, Levine J, Harris T, and others. Long-term functional outcome of dogs with severe injuries of the thoracolumbar spinal cord: 87 cases (1996-2001). J Am Vet Med Assoc. 2003; 222(6): 762-9. 17. Sharp NJH, Wheeler SJ. Small Animal Spinal Disorders. Diagnosis and Surgery. London. Elsevier 2005. 18. Allen GS, Wood AKW. Iohexol myelography in the dog. Vet Radiol Ultrasound 1988; 29: 78-82. 19. Butterworth SJ, Gibbs C. A review of the usefulness of myelography in 50 dogs Vet Rec 1992; 130 (21): 461-465. 20. Verstraete KL, Huysse WC. Health technology assessment of magnetic resonance imaging of the spine and bone marrow. Eur J Radiol 2008; 65(2): 201-10. 21. Ferreira AJA, Correia JHD, Jaggy A. Thoracolumbar disc disease in 71 paraplegic dogs: influence of rate of onset and duration of clinical signs on treatment results. J Small Anim Pract 2002; 43: 158–63. 22. Macias C, McKee WM, May C, Innes JF. Thoracolumbar disc disease in large dogs: a study of 99 cases J Sm Anim Pract 2002; 43: 439–446. 23. Prata RG. Neurosurgical treatment of thoracolumbar disks: the rationale and value of laminectomy and concomitant disk removal. J Am Anim Hosp Assoc 1981; 17:17–26. 24. Ruddle TL, Allen DA, Schertel ER and others. Outcome and prognostic factors in nonambulatory Hansen Type I intervertebral disc extrusions: 308 cases. Vet Comp Orthop Traumatol. 2006; 19: 29–34. 25. Moissonnier P, Meheust P, Carozzo C. Thoracolumbar lateral corpectomy for treatment of chronic disk herniation: technique description and use in 15 dogs. Vet Surg. 2004; 33(6): 620-8. 26. McKee, WM. Intervertebral disc disease in the dog: pathophysiology and diagnosis. In Pract 2000; 22: 355-369. 27. Chambers JN, Oliver JE Jr Kornegay JN, Malnati GA. Ventral decompression for caudal cervical disc herniation in large and giant breed dogs. J Am Vet Med Assoc 1982; 180: 410-414. 28. Lyman R. Continuous dorsal laminectomy is the procedure of choice. Prog Vet Neurol 1991; 2: 143-146. 29. Dixon BC, Tomlinson JL & Kraus KH. Modified distraction-stabilization technique using an interbody polymethyl methacrylate plug in dogs with caudal cervical spondylomyelopathy. J Am Vet Med Assoc 1996; 208: 61-68. 30. Ellison GW, Seim HB 3rd, Clemmons RM. Distracted cervical spinal fusion for management of caudal cervical spondylomyelopathy in large breed dogs. J Am Vet Med Assoc 1988; 193: 447-453. 31. Bergman RL, Levine JM, Coates JR, Bahr A, Hettlich BF, Kerwin SC. Cervical spinal locking plate in combination with cortical ring allograft for a one level fusion in dogs with cervical spondylotic myelopathy. Vet Surg 2008; 37: 530-536. 32. McKee WM, Sharp NJ. Cervical Spondylopathy. In: Textbook of Small Animal Surgery. 3rd edition. Slatter (ed.) Philadelphia: WB Saunders Co. 2003; 1180-1192. 33. Jeffery ND, McKee WM. Surgery for disc-associated wobbler syndrome in the dog – an examination of the controversy. J Small Anim Pract 2001; 42: 574-581. 34. Levine JM, Levine GJ, Johnson SI and others. Evaluation of the success of medical management for presumptive cervical intervertebral disk herniation in dogs. Vet Surg 2007; 36(5): 492-9. 35. Levine JM, Levine GJ, Johnson SI and others. Evaluation of the success of medical management for presumptive thoracolumbar intervertebral disk herniation in dogs. Vet Surg 2007; 36(5): 482-491. 36. Davis JV, Sharp NJH. A Comparison of conservative treatment and fenestration for thoracolumbar intervertebral disc disease in the dog. J Sm Anim Pract 1983; 24: 721-729. 37. Arias MJ. Treatment of experimental spinal cord injury with TRH, naloxone and dexamethasone. Surg Neurol 1987; 28: 335-338. 38. Kube SA, Olby NJ. Managing acute spinal cord injuries. Comp Cont Edu Vet 2008; 30(9): 496-506. 39. Moore RW, Withrow SJ. Gastrointestinal hemorrhage and pancreatitis associated with intervertebral disk diseases in the dog. J Am Vet Med Assoc 1982; 180(12): 1443-7. 40. LeCouter RA, Sturgess B Spinal Fractures and Luxations. In: Textbook of Small Animal Surgery. 3rd edn. Slatter DH (ed). Philadelphia: Saunders 2002; 1244-1260. 41. Levine JM, Levine GJ, Boozer L, and others. Adverse effects and outcome associated with dexamethasone administration in dogs with acute thoracolumbar intervertebral disk herniation: 161 cases (2000-2006). J Am Vet Med Assoc 2008; 232(3): 411-7. 42. Hall ED, Springer JE. Neuroprotection and acute spinal cord injury: a reappraisal. Neuro Rx 2004; 1: 80-100. 43. Hurlebert RJ. Methylprednisolone for acute spinal cord injury: an inappropriate standard of care. J Neurosurg 2000; 26: S39-46. 44. Vaquero J, Zurita M, Oya S, and others. Early administration of methylprednisolone decreases apoptotic cell death after spinal cord injury Histol Histopathol. 2006; 21(10): 1091-102. 45. Coates JR, Sorjonen DC, Simpson ST and others. Clinicopathologic effects of a 21-aminosteroid compound (U74389G) and high-Dose methylprednisolone on spinal cord function after simulated spinal cord trauma. Vet Surg. 1995; 24(2): 128-39. 46. Culbert LA, Marino DJ Baule RM, Knox III VW. Complications associated with high-dose prednisolone sodium succinate therapy in dogs with neurological injury. J Am Anim Hosp Assoc 1998; 34: 129–34. 47. Hanson SM, Bostwick DR, Twedt DC, Smith MO. Clinical evaluation of cimetidine, sucralfate, and misoprostol for prevention of gastrointestinal tract bleeding in dogs undergoing spinal surgery. Am J Vet Res 1997; 58(11): 1320-3. 48. Rohrer CR, Hill RC, Fischer A, and others. Gastric hemorrhage in dogs given high doses of methylprednisolone sodium succinate. Am J Vet Res. 1999; 60(8): 977-81. 49. Baptiste DC, Fehlings MG. Pharmacological approaches to repair the injured spinal cord. J Neurotrauma 2006; 23(3-4): 318-34 Review. 50. Spencer MT, Bazarian JJ. Are corticosteroids effective in traumatic spinal cord injury? Ann Emerg Med 2003; 41: 410-413. 51. Keilhoff G, Schild L, Fansa H. Minocycline protects Schwann cells from ischemia-like injury and promotes axonal outgrowth in bioartificial nerve grafts lacking Wallerian degeneration. Exp Neurol. 2008; 212(1): 189-200. 52. Gorio A, Gokmen N, Erbayraktar S, and others. Recombinant human erythropoietin counteracts secondary injury and markedly enhances neurological recovery from experimental spinal cord trauma. Proc Natl Acad Sci U S A. 2002; 99(14): 9450-5. 53. Laverty PH, Leskovar A, Breur GJ, and others. A preliminary study of intravenous surfactants in paraplegic dogs: polymer therapy in canine clinical SCI. J Neurotrauma. 2004; 21(12): 1767-77. 54. Syková E, Jendelová P. In vivo tracking of stem cells in brain and spinal cord injury. Prog Brain Res. 2007;161:367-83. Review. 55. Davis GJ, Brown DC. Prognostic indicators for time to ambulation after surgical decompression in nonambulatory dogs with acute thoracolumbar disc extrusions: 112 cases. Vet Surg 2002; 31: 513–8. 56. Jayaraman A, Shah P, Gregory C, and others. Locomotor training and muscle function after incomplete spinal cord injury: case series. J Spinal Cord Med. 2008; 31(2):185-93. 57. Behrman AL, Harkema SJ. Locomotor Training After Human Spinal Cord Injury: A Series of Case Studies Phys Ther 2000; 80(7): 688-700.
This article was first published in 2010.

